Key takeaways:
~ APOBEC3 is a family of enzymes that helps the body fight viral infections by causing mutations in viral DNA.
~ APOBEC3G specifically targets retroviruses by interfering with their reverse transcriptase.
~ APOBEC3-driven mutations can lead to viral variants and have been linked to about 22% of human viral species.
~ In cancer, APOBEC3 enzymes may contribute to mutations in cancer cells, particularly in virus-initiated cancers like cervical cancer.
~ Genetic variants in APOBEC3 affect viral response and the risk for certain types of cancer.
What is APOBEC3?
Throughout human history, we have constantly battled invisible pathogenic foes. Humans have sophisticated, built-in systems for fighting off viruses — this is how we have survived as a species.
One of the enzyme families involved in responding to viruses is called APOBEC3. It has a unique way of stopping viruses from replicating. Essentially, it causes mutations in the viral DNA, rendering the virus inactive. Once in a while, though, the mutations can increase the virulence of a virus.
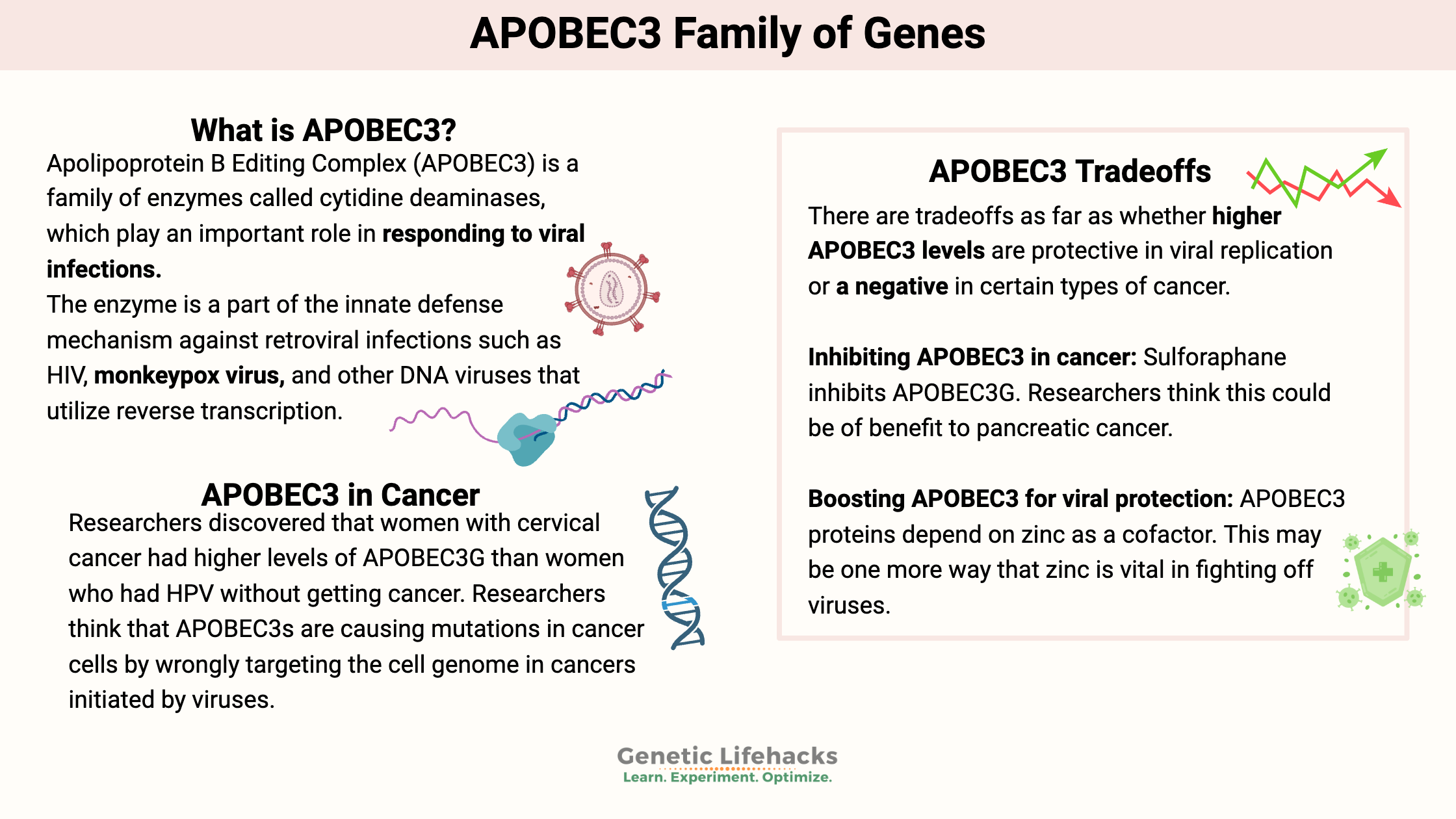
Apolipoprotein B Editing Complex (APOBEC3) is a family of enzymes called cytidine deaminases, which play an important role in responding to viral infections.
The APOBEC3G gene encodes an enzyme that is a part of the innate defense mechanism against retroviral infections as well as a few other types of viruses.
Retroviruses insert their viral RNA into the genome of the host cell’s DNA to be transcribed. While typical viruses use the host’s cellular mechanisms to convert viral RNA into proteins, retroviruses do things differently and use reverse transcriptase to produce DNA from the viral RNA. That DNA is then incorporated into the host cell’s genome. (While this sounds pretty wild, we actually have a lot of old viral DNA in our human genome.)
Once the retrovirus is incorporated into the host cell’s nuclear DNA, the virus proteins can be transcribed and translated, just like our regular genes are transcribed and translated. The viral proteins then can assemble new copies of the virus.
Retroviruses include HIV, but HIV has evolved the Viral infectivity factor protein (Vif), which counteracts the effects of APOBEC3G.
APOBEC3G is also part of the defense mechanism against poxviruses such as the monkeypox virus and other DNA viruses that utilize reverse transcription.
Viruses that are acted upon by by members of the APOBEC3 family include:[ref][ref][ref]
- Human T-lymphotropic virus
- herpes simplex 1
- RSV
- mumps
- HPV
- measles
- Epstein-Barr virus
The APOBEC3 family of proteins also inhibits coronavirus infections, although the mechanism of action there is not yet determined.[ref]
Some researchers think that APOBEC3-driven mutations are causing most of the variation in the SARS-CoV-2 virus.[ref]
What does APOBEC3 do?
APOBEC3G interferes with reverse transcriptase by causing mutations in the creation of the viral DNA. Specifically, it causes mutations converting G (guanine) to A (adenine) in the viral DNA. The mutations then may cause the virus to not function or not replicate well.[ref]
While the mutations in the viral DNA caused by APOBEC3G can cause the virus to no longer be infectious, it can also sometimes cause mutations that still allow the virus to function, just differently than before. In other words, APOBEC3G can cause variants in a virus.
Overall, researchers found that approximately 22% of human viral species are shaped by APOBEC3-causing mutations that result in positive evolutionary pressure in the virus.[ref]
In addition to causing hypermutations in retroviruses, researchers think that APOBEC3G also inhibits viral replication by other means. Cell studies show that APOBEC3G can inhibit infection by parvoviruses without hypermutating their genomes.[ref]
Interferon is one of the first innate immune system signals produced in a viral infection. APOBEC3G is upregulated in response to interferon. It is also produced in T cells, B cells, and macrophages, and B cells can release it in exosomes.[ref]
APOBEC3 in cancer:
HPV (human papillomavirus) is the primary cause of cervical cancer. While most HPV infections are harmless and clear up spontaneously, persistent HPV infections can lead to cancer in certain circumstances. Recently, researchers discovered that women with cervical cancer had higher levels of APOBEC3G than women who had HPV without getting cancer.[ref]
Researchers think that APOBEC3s are causing mutations in cancer cells by wrongly targeting the cell genome in cancers initiated by viruses. One study explains: “For example, high-risk papillomaviruses, whose viral genomes carry an APOBEC3 footprint, indirectly promote cell transformation due to the sustained APOBEC3 mutagenic activity.”[ref]
APOBEC3 Genotype Report:
Researchers have found that the APOBEC3 family of genes has diverse variants in population groups worldwide. It may be because specific variants have been selected by combating endemic viruses in certain parts of the world.[ref]
Unfortunately, only a few of these variants are covered by 23andme or AncestryDNA data.
Lifehacks:
There are tradeoffs as far as whether higher APOBEC3 levels are protective in viral replication or a negative in certain types of cancer. Keep in mind that not all cancers are the same, and always talk with your doctor if you have any questions about whether a supplement is right for you.
Related Articles and Topics:
References:
An, Ping, et al. “Role of APOBEC3F Gene Variation in HIV-1 Disease Progression and Pneumocystis Pneumonia.” PLoS Genetics, vol. 12, no. 3, Mar. 2016, p. e1005921. PubMed, https://doi.org/10.1371/journal.pgen.1005921.
Bogdanova, Natalia, et al. “Hereditary Breast Cancer: Ever More Pieces to the Polygenic Puzzle.” Hereditary Cancer in Clinical Practice, vol. 11, no. 1, Sept. 2013, p. 12. PubMed Central, https://doi.org/10.1186/1897-4287-11-12.
Chen, Zhishan, et al. “Integrative Genomic Analyses of APOBEC-Mutational Signature, Expression and Germline Deletion of APOBEC3 Genes, and Immunogenicity in Multiple Cancer Types.” BMC Medical Genomics, vol. 12, Sept. 2019, p. 131. PubMed Central, https://doi.org/10.1186/s12920-019-0579-3.
Compaore, Tegwinde Rebeca, et al. “APOBEC3G Variants and Protection against HIV-1 Infection in Burkina Faso.” PloS One, vol. 11, no. 1, 2016, p. e0146386. PubMed, https://doi.org/10.1371/journal.pone.0146386.
He, Xiu-Ting, et al. “Association between Polymorphisms of the APOBEC3G Gene and Chronic Hepatitis B Viral Infection and Hepatitis B Virus-Related Hepatocellular Carcinoma.” World Journal of Gastroenterology, vol. 23, no. 2, Jan. 2017, pp. 232–41. PubMed, https://doi.org/10.3748/wjg.v23.i2.232.
Iqbal, Khurshid, et al. “Correlation of Apolipoprotein B MRNA-Editing Enzyme, Catalytic Polypeptide-like 3G Genetic Variant Rs8177832 with HIV-1 Predisposition in Pakistani Population.” Current HIV Research, vol. 16, no. 4, July 2018, pp. 297–301. PubMed Central, https://doi.org/10.2174/1570162X16666181018155827.
Luo, Yiqiao, et al. “Sulforaphane Inhibits the Expression of Long Noncoding RNA H19 and Its Target APOBEC3G and Thereby Pancreatic Cancer Progression.” Cancers, vol. 13, no. 4, Feb. 2021, p. 827. PubMed Central, https://doi.org/10.3390/cancers13040827.
Mourier, Tobias, et al. “Host-Directed Editing of the SARS-CoV-2 Genome.” Biochemical and Biophysical Research Communications, vol. 538, Jan. 2021, pp. 35–39. PubMed Central, https://doi.org/10.1016/j.bbrc.2020.10.092.
Park, Charny, et al. “Integrative Molecular Profiling Identifies a Novel Cluster of Estrogen Receptor-Positive Breast Cancer in Very Young Women.” Cancer Science, vol. 110, no. 5, May 2019, pp. 1760–70. PubMed, https://doi.org/10.1111/cas.13982.
Poulain, Florian, et al. “Footprint of the Host Restriction Factors APOBEC3 on the Genome of Human Viruses.” PLOS Pathogens, vol. 16, no. 8, Aug. 2020, p. e1008718. PLoS Journals, https://doi.org/10.1371/journal.ppat.1008718.
Sadeghpour, Shiva, et al. “Human APOBEC3 Variations and Viral Infection.” Viruses, vol. 13, no. 7, July 2021, p. 1366. PubMed Central, https://doi.org/10.3390/v13071366.
Sadler, Holly A., et al. “APOBEC3G Contributes to HIV-1 Variation through Sublethal Mutagenesis.” Journal of Virology, vol. 84, no. 14, July 2010, pp. 7396–404. PubMed Central, https://doi.org/10.1128/JVI.00056-10.
Sharma, Shraddha, et al. “APOBEC3A Cytidine Deaminase Induces RNA Editing in Monocytes and Macrophages.” Nature Communications, vol. 6, Apr. 2015, p. 6881. PubMed Central, https://doi.org/10.1038/ncomms7881.
Stavrou, Spyridon, and Susan R. Ross. “APOBEC3 Proteins in Viral Immunity.” Journal of Immunology (Baltimore, Md. : 1950), vol. 195, no. 10, Nov. 2015, pp. 4565–70. PubMed Central, https://doi.org/10.4049/jimmunol.1501504.
Sui, Shuang, et al. “Correlation of APOBEC3G Polymorphism with Human Papillomavirus (HPV) Persistent Infection and Progression of Cervical Lesions.” Medical Science Monitor : International Medical Journal of Experimental and Clinical Research, vol. 25, Sept. 2019, pp. 6990–97. PubMed Central, https://doi.org/10.12659/MSM.916142.