Key Takeaways:
~ Gilbert’s syndrome is caused by a mutation in a gene that affects the liver’s ability to break down bilirubin, which is a waste product from old red blood cells.
~ Slightly elevated bilirubin usually causes no issues. But for some, there are symptoms due to specific conditions.
~ Gilbert’s syndrome is caused by changes in the UGT1A1 gene.
This article dives into the genetics of Gilbert’s syndrome, along with some fascinating science on bilirubin. I’ll explain what bilirubin does in the body, how Gilbert’s syndrome is actually beneficial in some ways, and what to watch out for.
Members will see their genotype report below, plus additional solutions in the Lifehacks section. Join today.
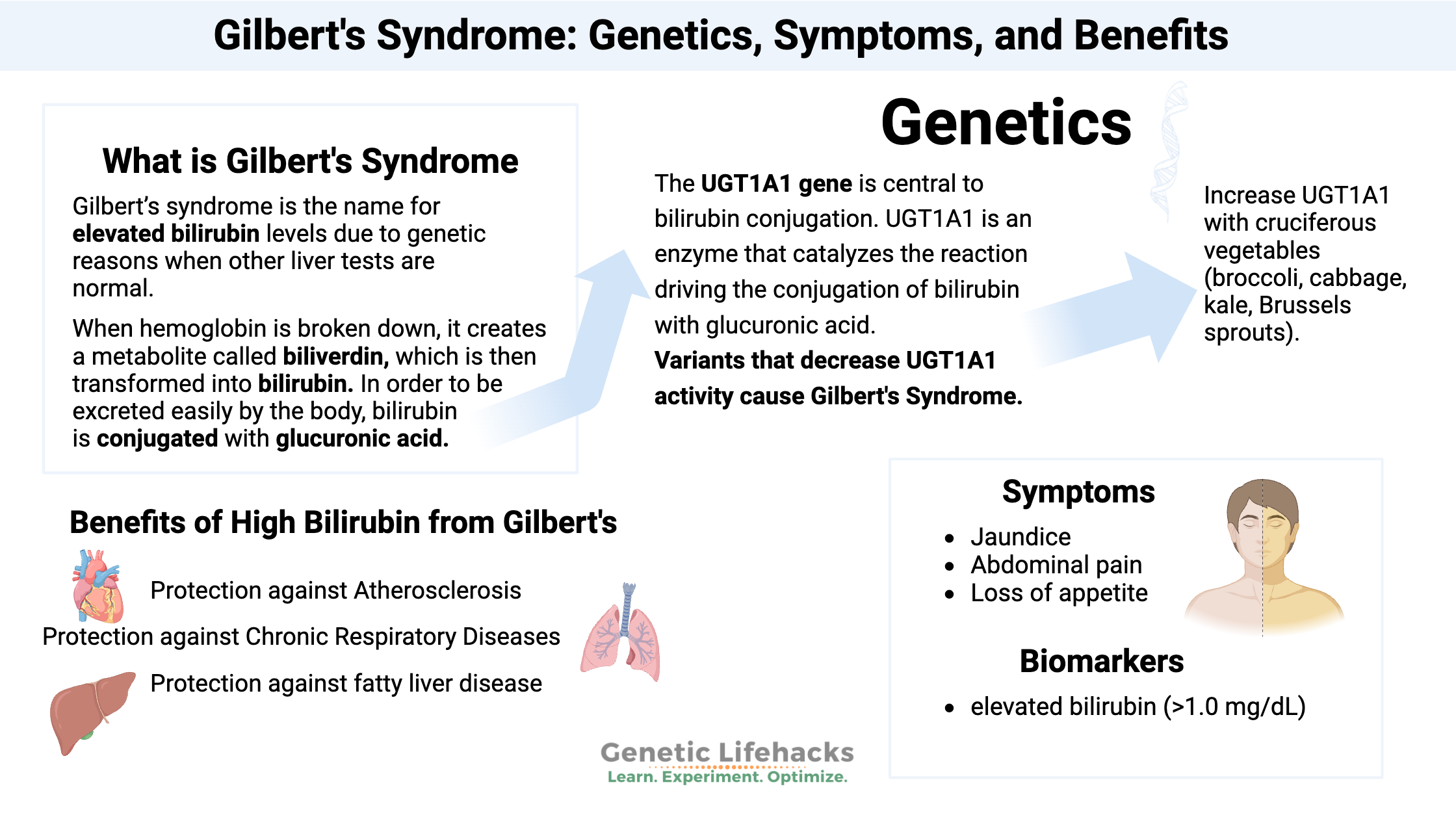
What is Gilbert’s Syndrome?
Gilbert’s syndrome – pronounced Zheel-bayr syndrome – is the name for elevated bilirubin levels due to genetic reasons. Gilbert’s syndrome is somewhat common and is often diagnosed through routine blood tests. It affects about 5-10% of the population.[ref]
The mild elevation in bilirubin usually has no detrimental effects, but mild jaundice (yellowing of the skin and eyes) may appear under certain conditions. For people with Gilbert’s syndrome, bilirubin levels may be higher when fasting, after strenuous exercise, when stressed out, or during an illness.
Gilbert’s syndrome is diagnosed by doctors when bilirubin alone is elevated, and other liver tests are in the normal range. In other words, if bilirubin is high and other liver enzymes are out of range, then elevated bilirubin can indicate jaundice from liver disease.
Let’s dig into the details first on what bilirubin does in the body.
What is bilirubin?
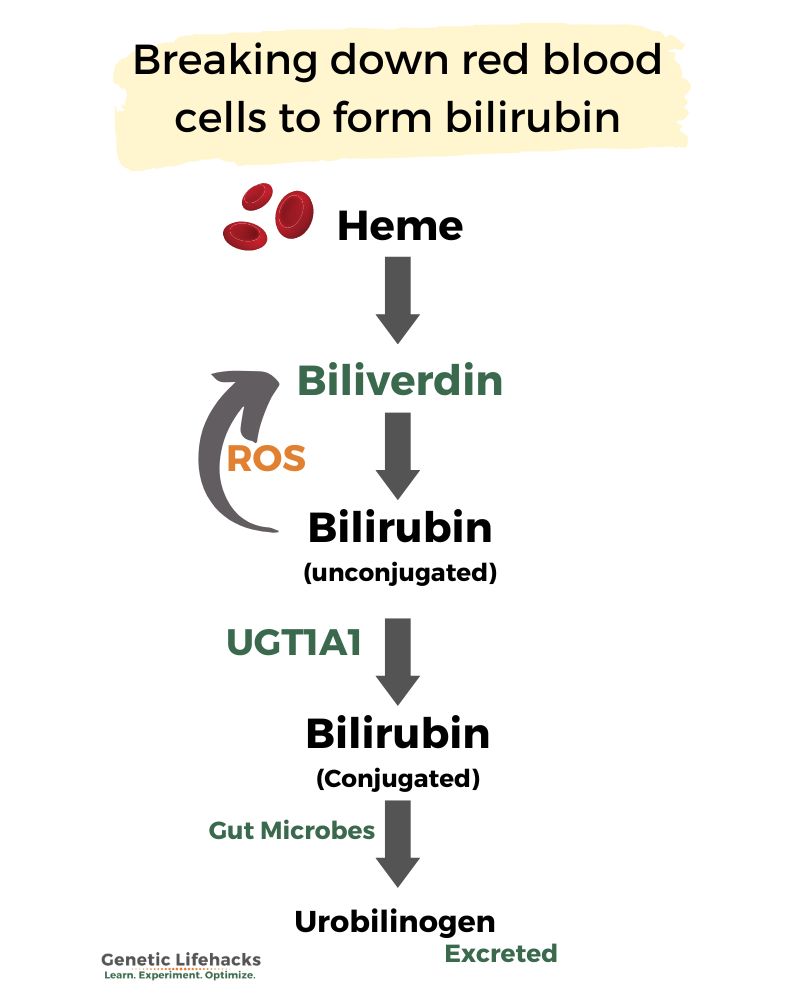
Bilirubin is produced during the breakdown of red blood cells.
The body constantly produces new red blood cells and breaks down old ones. In fact, the average lifespan of a red blood cell is only four months, after which they are broken down and recycled.
A type of white blood cell called a macrophage recognizes certain marks on aging red blood cells and engulfs them to clear them out. In the bone marrow, new red blood cells are constantly being formed to replace the old cells. Old red blood cells are primarily removed and ingested by macrophages in the spleen.[ref]
Hemoglobin is the protein that carries oxygen in red blood cells. When hemoglobin is broken down, it creates a metabolite called biliverdin. The biliverdin is then transformed into bilirubin.[ref]
hemoglobin -> bilverdin -> bilirubin
Bilirubin is a reddish-orange colored compound. This color is what shows up as yellow in jaundice and healing bruises. It is also part of why feces is brown.
In order to be excreted easily by the body, bilirubin is conjugated (bound together) with glucuronic acid. This binding makes the conjugated bilirubin water soluble and able to be excreted in bile through the intestines.
Unconjugated bilirubin can’t be easily excreted and instead will circulate in the bloodstream.
Gilbert’s syndrome is an elevation of unconjugated bilirubin due to a lack of the UGT1A1 enzyme needed in the conjugation reaction.
UGT1A1 enzyme: conjugating bilirubin
The UGT1A1 gene is central to bilirubin conjugation. UGT1A1 is uridine diphosphoglucuronyltransferase 1, which is an enzyme that catalyzes the reaction driving the conjugation of bilirubin with glucuronic acid.[ref]
Essentially, unconjugated bilirubin can take one of two paths:
- It can convert back to biliverdin by acting as an antioxidant in the presence of reactive oxygen species (ROS)
– OR – - It can be conjugated with glucuronic acid and then excreted in feces.
The first path – acting as an antioxidant – can become important when cells produce a lot of reactive oxygen species, such as in heart disease. And this is where the benefits of low UGT1A1, and thus more bilirubin, come into play. More on this below in the benefits of Gilbert’s syndrome section.
With plenty of UGT1A1 available, more bilirubin will be conjugated and excreted.
UV light, bilirubin, and jaundice in babies:
Bilirubin is a pigment, and, like other pigments, light can affect its chemical bonds. The double bonds in bilirubin can isomerize when exposed to light, which results in the bilirubin becoming more soluble.
In babies with jaundice, UV lights are used to help the bilirubin to be more soluble and more easily excreted. Just after birth, babies don’t produce UGT1A1.
While jaundice due to Gilbert’s syndrome isn’t much of a problem in adults or kids, unconjugated bilirubin at high levels in newborns can cause the bilirubin to accumulate in some regions of the brain, causing damage.
Bilirubin can get to infants’ brains because the blood-brain barrier may not yet be fully developed.[ref] Infants also don’t have a fully developed gut microbiome yet, so the impact of intestinal bacteria on bilirubin is different in babies (and also why their poop is lighter colored).
This neatly brings us to the topic of what the gut microbiome is doing with bilirubin…
Gut microbiome and bilirubin:
Once bilirubin is conjugated using the UGT1A1 enzyme, it is released into the intestines from the liver via bile. The bacteria in the gut microbiome then de-conjugate the glucuronic acid from the bilirubin. The bilirubin is then oxidized and reduced, eventually forming stercobilin (excreted in the feces) or urobilin (reabsorbed and excreted in the urine).[ref]
The deconjugation reaction is facilitated by β-glucuronidase, an enzyme produced by gut bacteria. Thus, the gut microbiome affects how bilirubin is excreted via urine or feces.
Jaundice and Symptoms of Gilbert’s Syndrome:
While many people are diagnosed with Gilbert’s syndrome through routine blood tests, others may have symptoms that prompt them to go to the doctor.
In a survey of patients diagnosed with Gilbert’s syndrome, about half of them had recurrent jaundice. About half of those with recurrent jaundice reported abdominal pain, dyspepsia, or loss of appetite.
Risk factors associated with causing the jaundice episodes included:[ref]
- fasting for more than 12 hours
- pregnancy
- low-calorie diets
- infection
- anesthesia
- intense physical activity
A Genetic Lifehacks member with Gilbert’s Syndrome explained the symptoms to me:
“… sometimes the symptoms get in the way of living. For example, if I go more than twelve hours without food, I have to plan for an hour or two of fatigue until I recover. Likewise, if I get sick, I’m usually down for twice as long as anyone else. Strenuous exercise is totally out — 3-day recovery. I don’t often turn yellow, but, it really sucks when I do. Also, it helps if I eat something every 3-4 hours, and get plenty of protein.”
Benefits of higher bilirubin levels in Gilbert’s Syndrome:
When bilirubin is elevated due to genetics, there are several noted benefits. These benefits are derived from bilirubin acting as an antioxidant, neutralizing excess ROS. Many of the benefits are seen more in older adults, around age 50, when the toll of excess oxidative stress is likely to cause many of the problems of aging.[ref]
Protection against Atherosclerosis:
The most significant benefit of higher bilirubin is a decrease in atherosclerotic plaque buildup. As you’ll see in the genotype report, the variants linked to higher bilirubin are also linked to a lower risk of heart disease.
Protection against Chronic Respiratory Diseases:
A UK study looked at Gilbert’s syndrome’s effect on respiratory disease risk in people born in 1946. Many people in this cohort had smoked, and those with Gilbert’s syndrome were less likely to have COPD or asthma than those with normal bilirubin.[ref]
Protection against fatty liver disease:
Animal studies show that the UGT1A1 variants that increase bilirubin also decrease the risk and severity of fatty liver disease (NAFLD).[ref] A meta-analysis including more than 100,000 participants found that higher bilirubin levels are associated with lower odds of NAFLD.[ref]
Overall decreased mortality:
A research study with over 4200 people with Gilbert’s syndrome and ~22,000 with normal bilirubin found that the mortality rate was decreased in Gilbert’s syndrome. The results showed the adjusted mortality rate was about 50% less in people with Gilbert’s syndrome.[ref]
Thus, the drawback of possibly having jaundice during times of physical stress is offset, for some people, with a reduced risk of cardiovascular disease, COPD, or liver disease.
Gilbert’s Syndrome Genotype Report:
Lifehacks:
Testing: If you are concerned, talk with your doctor about getting your bilirubin tested. You can also order lab tests on your own in the US. Bilirubin is often included in the CBC panel, which is very inexpensive.
Lifestyle Considerations with Gilbert’s Syndrome:
For people with reduced UGT1A1 function, bilirubin may be elevated to the point of having jaundice (and possibly abdominal pain) by activities that increase the breakdown of red blood cells. Examples include:
- pregnancy
- illness
- fasting
- dehydration
- excessive exercise (e.g., long-distance running)
- anesthesia or surgery
- stressful low-calorie dieting
It doesn’t mean you can’t exercise, go on a diet, or get pregnant due to Gilbert’s syndrome. Rather, take this as a ‘heads up’ that you may have elevated bilirubin and perhaps jaundice in times of physical stress.
Take care of your liver by eating well, avoiding excessive alcohol, and cutting down on high fructose corn syrup (leading to NAFLD).
According to the Cleveland Clinic, jaundice in Gilbert’s syndrome doesn’t pose a health risk.[ref] Check with your doctor if you have any questions or changes in skin color or urine color. Elevated bilirubin could be due to other liver problems, in addition to Gilbert’s Syndrome.
Chemotherapy:
If you have Gilbert’s syndrome or the UGT1A1 variants above, it is important to talk with your oncologist about it if you are undergoing chemotherapy. Certain chemo drugs are metabolized using the UGT1A1 enzyme.
Toxicants that need UGT1A1 for metabolism:
Related Articles and Topics:
Glucuronidation: UGT Genetic Variants, Detoxification, and Hormone Balance
References:
Bale, Govardhan, et al. “Incidence and Risk of Gallstone Disease in Gilbert’s Syndrome Patients in Indian Population.” Journal of Clinical and Experimental Hepatology, vol. 8, no. 4, Dec. 2018, pp. 362–66. PubMed Central, https://doi.org/10.1016/j.jceh.2017.12.006.
Buch, Stephan, et al. “Loci from a Genome-Wide Analysis of Bilirubin Levels Are Associated with Gallstone Risk and Composition.” Gastroenterology, vol. 139, no. 6, Dec. 2010, pp. 1942-1951.e2. PubMed, https://doi.org/10.1053/j.gastro.2010.09.003.
Chen, Guanjie, et al. “A UGT1A1 Variant Is Associated with Serum Total Bilirubin Levels, Which Are Causal for Hypertension in African-Ancestry Individuals.” NPJ Genomic Medicine, vol. 6, no. 1, June 2021, p. 44. PubMed, https://doi.org/10.1038/s41525-021-00208-6.
Franco, Marco E., et al. “Altered Expression and Activity of Phase I and II Biotransformation Enzymes in Human Liver Cells by Perfluorooctanoate (PFOA) and Perfluorooctane Sulfonate (PFOS).” Toxicology, vol. 430, Jan. 2020, p. 152339. PubMed, https://doi.org/10.1016/j.tox.2019.152339.
“Gilbert’s Syndrome: Symptoms, Causes, Tests & Treatment.” Cleveland Clinic, https://my.clevelandclinic.org/health/diseases/17661-gilberts-syndrome. Accessed 1 Sept. 2022.
Ha, Vincent H., et al. “Oncology Drug Dosing in Gilbert Syndrome Associated with UGT1A1: A Summary of the Literature.” Pharmacotherapy, vol. 37, no. 8, Aug. 2017, pp. 956–72. PubMed, https://doi.org/10.1002/phar.1946.
Hansen, Thor W. R., et al. “Molecular Physiology and Pathophysiology of Bilirubin Handling by the Blood, Liver, Intestine, and Brain in the Newborn.” Physiological Reviews, vol. 100, no. 3, July 2020, pp. 1291–346. PubMed, https://doi.org/10.1152/physrev.00004.2019.
Horsfall, Laura J., Rebecca Hardy, et al. “Genetic Variation Underlying Common Hereditary Hyperbilirubinaemia (Gilbert’s Syndrome) and Respiratory Health in the 1946 British Birth Cohort.” Journal of Hepatology, vol. 61, no. 6, Dec. 2014, pp. 1344–51. PubMed, https://doi.org/10.1016/j.jhep.2014.07.028.
Horsfall, Laura J., Irwin Nazareth, et al. “Gilbert’s Syndrome and the Risk of Death: A Population-Based Cohort Study.” Journal of Gastroenterology and Hepatology, vol. 28, no. 10, Oct. 2013, pp. 1643–47. PubMed, https://doi.org/10.1111/jgh.12279.
Kamal, Sanaa, et al. “The Frequency, Clinical Course, and Health Related Quality of Life in Adults with Gilbert’s Syndrome: A Longitudinal Study.” BMC Gastroenterology, vol. 19, Feb. 2019, p. 22. PubMed Central, https://doi.org/10.1186/s12876-019-0931-2.
Konaka, Ken, et al. “Study on the Optimal Dose of Irinotecan for Patients with Heterozygous Uridine Diphosphate-Glucuronosyltransferase 1A1 (UGT1A1).” Biological & Pharmaceutical Bulletin, vol. 42, no. 11, 2019, pp. 1839–45. PubMed, https://doi.org/10.1248/bpb.b19-00357.
Kuo, Sung-Hsin, et al. “Polymorphisms of ESR1, UGT1A1, HCN1, MAP3K1 and CYP2B6 Are Associated with the Prognosis of Hormone Receptor-Positive Early Breast Cancer.” Oncotarget, vol. 8, no. 13, Mar. 2017, pp. 20925–38. PubMed, https://doi.org/10.18632/oncotarget.14995.
Landerer, Steffen, et al. “A Gilbert Syndrome-Associated Haplotype Protects against Fatty Liver Disease in Humanized Transgenic Mice.” Scientific Reports, vol. 10, May 2020, p. 8689. PubMed Central, https://doi.org/10.1038/s41598-020-65481-4.
Liang, Chen, et al. “Association of Serum Bilirubin With Metabolic Syndrome and Non-Alcoholic Fatty Liver Disease: A Systematic Review and Meta-Analysis.” Frontiers in Endocrinology, vol. 13, July 2022, p. 869579. PubMed Central, https://doi.org/10.3389/fendo.2022.869579.
Lv, Xia, et al. “Recent Progress and Challenges in Screening and Characterization of UGT1A1 Inhibitors.” Acta Pharmaceutica Sinica. B, vol. 9, no. 2, Mar. 2019, pp. 258–78. PubMed Central, https://doi.org/10.1016/j.apsb.2018.09.005.
Maekawa, Shinya, et al. “Association between Alanine Aminotransferase Elevation and UGT1A1*6 Polymorphisms in Daclatasvir and Asunaprevir Combination Therapy for Chronic Hepatitis C.” Journal of Gastroenterology, vol. 53, no. 6, June 2018, pp. 780–86. PubMed, https://doi.org/10.1007/s00535-017-1405-3.
Oussalah, Abderrahim, et al. “Exome-Wide Association Study Identifies New Low-Frequency and Rare UGT1A1 Coding Variants and UGT1A6 Coding Variants Influencing Serum Bilirubin in Elderly Subjects: A Strobe Compliant Article.” Medicine, vol. 94, no. 22, June 2015, p. e925. PubMed, https://doi.org/10.1097/MD.0000000000000925.
Pang, Hui-Lin, et al. “Discovery and Characterization of the Key Constituents in Ginkgo Biloba Leaf Extract With Potent Inhibitory Effects on Human UDP-Glucuronosyltransferase 1A1.” Frontiers in Pharmacology, vol. 13, 2022. Frontiers, https://www.frontiersin.org/articles/10.3389/fphar.2022.815235.
Pentón-Rol, Giselle, et al. “C-Phycocyanin-Derived Phycocyanobilin as a Potential Nutraceutical Approach for Major Neurodegenerative Disorders and COVID-19-Induced Damage to the Nervous System.” Current Neuropharmacology, vol. 19, no. 12, Dec. 2021, pp. 2250–75. PubMed Central, https://doi.org/10.2174/1570159X19666210408123807.
Poblete, Daniela, et al. “Pharmacogenetic Associations Between Atazanavir/UGT1A1*28 and Efavirenz/Rs3745274 (CYP2B6) Account for Specific Adverse Reactions in Chilean Patients Undergoing Antiretroviral Therapy.” Frontiers in Pharmacology, vol. 12, 2021, p. 660965. PubMed, https://doi.org/10.3389/fphar.2021.660965.
Rs4124874 – SNPedia. https://snpedia.com/index.php/Rs4124874. Accessed 1 Sept. 2022.
Saito, Akira, et al. “Association Study between Single-Nucleotide Polymorphisms in 199 Drug-Related Genes and Commonly Measured Quantitative Traits of 752 Healthy Japanese Subjects.” Journal of Human Genetics, vol. 54, no. 6, June 2009, pp. 317–23. PubMed, https://doi.org/10.1038/jhg.2009.31.
Thiagarajan, Perumal, et al. “How Do Red Blood Cells Die?” Frontiers in Physiology, vol. 12, 2021. Frontiers, https://www.frontiersin.org/articles/10.3389/fphys.2021.655393.
TSUNEDOMI, RYOUICHI, et al. “A Novel System for Predicting the Toxicity of Irinotecan Based on Statistical Pattern Recognition with UGT1A Genotypes.” International Journal of Oncology, vol. 45, no. 4, July 2014, pp. 1381–90. PubMed Central, https://doi.org/10.3892/ijo.2014.2556.
Wagner, Karl-Heinz, et al. “Oxidative Stress and Related Biomarkers in Gilbert’s Syndrome: A Secondary Analysis of Two Case-Control Studies.” Antioxidants (Basel, Switzerland), vol. 10, no. 9, Sept. 2021, p. 1474. PubMed, https://doi.org/10.3390/antiox10091474.
Yang, Hui, et al. “UGT1A1 Mutation Association with Increased Bilirubin Levels and Severity of Unconjugated Hyperbilirubinemia in ABO Incompatible Newborns of China.” BMC Pediatrics, vol. 21, no. 1, June 2021, p. 259. PubMed, https://doi.org/10.1186/s12887-021-02726-9.
Yoda, Emiko, et al. “Isothiocyanates Induce UGT1A1 in Humanized UGT1 Mice in a CAR Dependent Fashion That Is Highly Dependent upon Oxidative Stress.” Scientific Reports, vol. 7, Apr. 2017, p. 46489. PubMed Central, https://doi.org/10.1038/srep46489.
Zanussi, Jacy T., et al. “Identifying Potential Therapeutic Applications and Diagnostic Harms of Increased Bilirubin Concentrations: A Clinical and Genetic Approach.” Clinical Pharmacology and Therapeutics, vol. 111, no. 2, Feb. 2022, pp. 435–43. PubMed, https://doi.org/10.1002/cpt.2441.
Zhou, Youyou, et al. “Association of UGT1A1 Variants and Hyperbilirubinemia in Breast-Fed Full-Term Chinese Infants.” PLoS ONE, vol. 9, no. 8, Aug. 2014, p. e104251. PubMed Central, https://doi.org/10.1371/journal.pone.0104251.
Zhu, Ya-Di, et al. “Neobavaisoflavone Induces Bilirubin Metabolizing Enzyme UGT1A1 via PPARα and PPARγ.” Frontiers in Pharmacology, vol. 11, 2021. Frontiers, https://www.frontiersin.org/articles/10.3389/fphar.2020.628314.
Zöhrer, Patrick A., et al. “Gilbert’s Syndrome and the Gut Microbiota – Insights From the Case-Control BILIHEALTH Study.” Frontiers in Cellular and Infection Microbiology, vol. 11, Sept. 2021, p. 701109. PubMed Central, https://doi.org/10.3389/fcimb.2021.701109.