Key takeaways:
~ Vitamin B12 is a cofactor for biological reactions such as creating the myelin sheath in nerve cells and the synthesis of DNA.
~ A lack of vitamin B12 (also known as cobalamin) can create a cascade of negative effects.
~ There are several genes that can influence how much vitamin B12 you absorb, transport, and need. Some people need higher amounts of B12, while others thrive on different forms of B12. Looking at your genetic data may help you figure out what is going on in your body.
Vitamin B12: Why we need it, where we get it, and what it does
Vitamin B12, also known as cobalamin, is an essential nutrient that is needed for DNA synthesis, red blood cell formation, nerve function, cellular energy, and homocysteine metabolism.
Foods that are high in vitamin B12 include meat, fish, eggs, and dairy. Vegetarian and vegan diets are lacking in vitamin B12, and supplementation is usually recommended.
Vitamin B12 as a supplement can be found in four different forms:
- cyanocobalamin (common in cheaper supplements)
- methylcobalamin (methylB12)
- adenosylcobalamin (adenosylB12)
- hydroxocobalamin (hydroxyB12)
The cyanocobalamin form is often found in cheaper vitamins and added to processed foods. It must be converted by the body before use. Methylcobalamin and adenosylcobalamin are active forms used by the body.
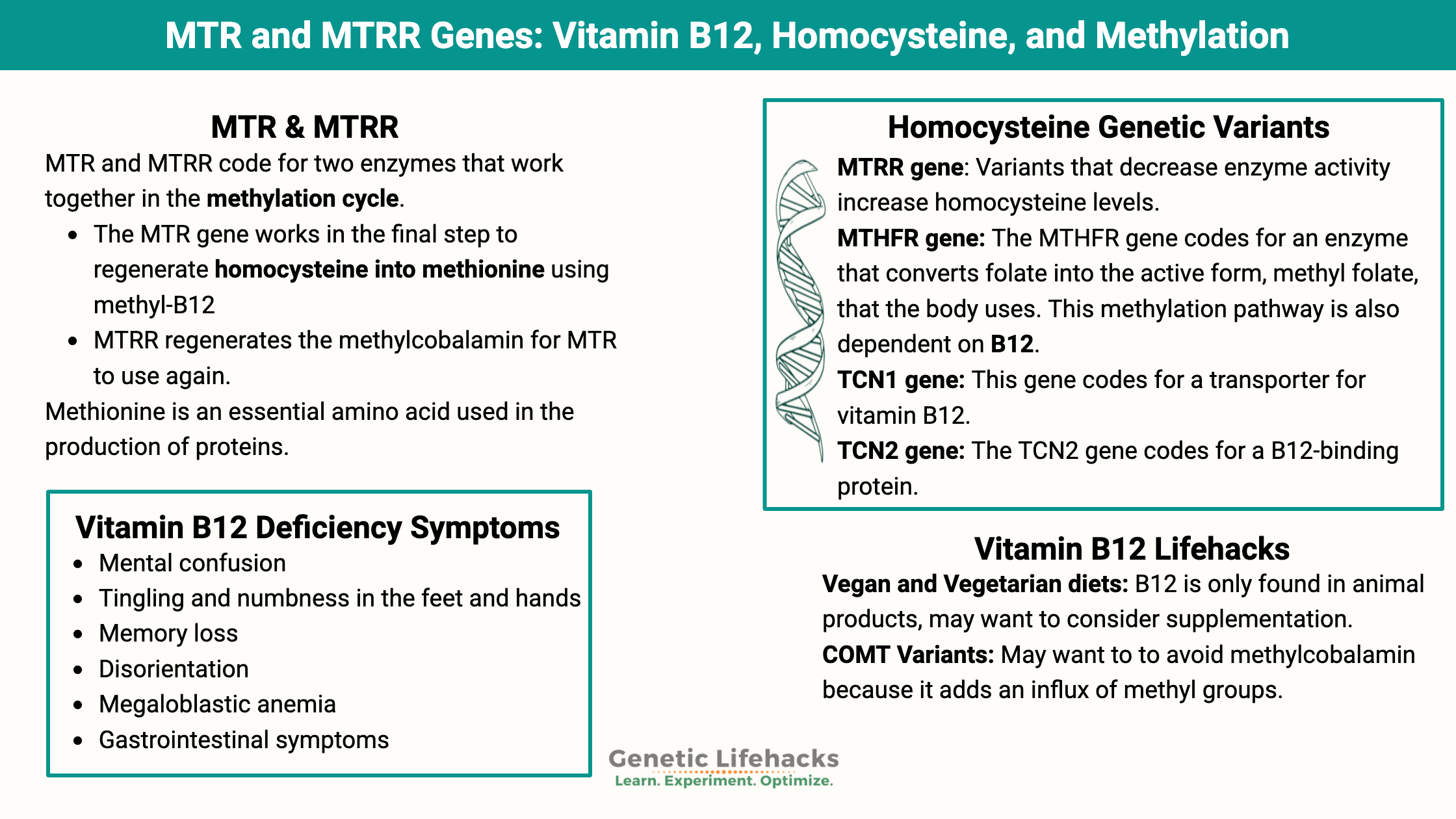
Vitamin B12 deficiency symptoms:
Vitamin B12 deficiency or insufficiency has been shown to cause:[ref][ref]
- mental confusion
- tingling and numbness in the feet and hands
- muscle cramps, nighttime leg cramps
- memory loss
- disorientation
- megaloblastic anemia
- gastrointestinal symptoms
To be able to absorb B12 from foods, you need to have adequate intrinsic factor produced in the stomach. This is something that is often depleted in the elderly, leading to B12 deficiency.
MTR & MTRR: Genetic connections between Methionine and Vitamin B12
Methionine is an essential amino acid used in the production of proteins. It is literally the starting amino acid for every protein your body makes.
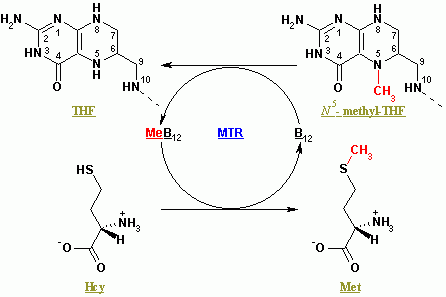
MTR (methionine synthase) and MTRR (methionine synthase reductase) code for two enzymes that work together in the methylation cycle.
- The MTR gene works in the final step to regenerate homocysteine into methionine using methyl-B12 (methylcobalamin)
- MTRR regenerates the methylcobalamin for MTR to use again.[ref]
Both are a vital part of the methylation cycle.
Methyl groups and methylation cycle: Quick background
A methyl group is made up of a carbon plus three hydrogen atoms. Your body is made up of organic molecules, a lot of which contain carbon bonded to hydrogen. Adding in a methyl group is like adding a building block to the molecule.
The methylation cycle is your body’s way of recycling certain molecules to ensure that there are enough methyl groups (carbon plus three hydrogens) available for cellular processes. When it comes to the functioning of your cells, methyl groups are used in methylation reactions.[ref]
Examples of methylation reactions include:
- synthesis of some of the nucleic acid (DNA) bases
- turning off genes so that they aren’t transcribed (DNA methylation)
- converting serotonin into melatonin
- methylating arsenic so that it can be excreted
- breaking down neurotransmitters
- metabolizing estrogen
- regenerating methionine from homocysteine
Methylation in the right amount:
For example, you need enough folate (vitamin B9) and methylcobalamin (vitamin B12) for the methionine synthase reaction to occur. Methylfolate is the source of the methyl group that methionine synthase uses for converting homocysteine to methionine. (Read more about your MTHFR genes and methyl folate)
Not enough B12 or methyl folate?
MTR won’t convert as much homocysteine to methionine, leading to a buildup of homocysteine and limiting methionine. Too much homocysteine is strongly associated with an increased risk of heart disease.[ref]
The other side of the picture, though, is that there may be times when limiting methionine is helpful, such as in fighting the proliferation of cancer cells. Excess vitamin B12 is linked to increased cancer growth in certain types of cancer.[ref]
Methotrexate, a chemotherapy drug, works by inhibiting the production of methyl folate, thus limiting methionine and DNA synthesis for cell growth.
TCN1 and TCN2: Transporting vitamin B12
TCN1 (transcobalamin I) is a protein that binds to and transports vitamin B12. It plays a role in cobalamin transport and metabolism, particularly in the stomach and intestines, where it protects B12 from being destroyed by stomach acid.
TCN2 (transcobalamin) is a B12 transport protein that moves vitamin B12 from the bloodstream into cells throughout the body. Rare mutations in TCN2 cause transcobalamin deficiency, which has symptoms of neurological problems, failure to thrive, vomiting, and low blood cell counts. Common genetic variants cause more minor changes in cellular B12 levels.[ref]
Vitamin B12 Genotype Report:
Lifehacks: Dietary options, picking the right B12 supplements
Dietary considerations:
Vegan and Vegetarian diets:
Vitamin B12 is only found in animal-based foods, so vegans and vegetarians are often deficient or marginal in their B12 status.[ref] B12 is often added to breakfast cereals and other refined products, so eating a vegetarian diet that includes packaged and refined foods may actually result in higher B12 levels (although probably not in better health).
It takes several years to completely deplete your liver’s stores of B12, so people who have recently started a vegan or vegetarian diet may still have moderate vitamin B12 levels.[ref]
Dietary sources:
Foods that are highest in B12 include:[ref]
- beef liver
- mussels
- lamb
- caviar
- chicken livers
High Homocysteine and B12:
Homocysteine levels:
If your homocysteine levels are high and you carry the MTHFR or MTRR variant reduced function variant above, supplementing with vitamin B12, methylfolate, riboflavin, and B6 may help to lower your levels.[ref][ref][ref]
Interaction with alcohol:
Alcohol is metabolized into acetaldehyde, which is toxic and inhibits methionine synthase (MTR). This is one reason why alcoholism causes B12 deficiency. If you have reduced MTR or MTRR activity, you may want to avoid excess acetaldehyde by not drinking much alcohol. [ref]
The Right form of Supplemental B12:
Related Articles and Topics:
How Well Do You Convert Beta-Carotene to Vitamin A?
Genetics plays a huge role in how well you convert beta-carotene into vitamin A. This article covers the research on the conversion of beta-carotene to vitamin A and how genetic variants decrease the conversion process for some people.
Should you take folic acid?
There is a lot of buzz online about MTHFR variants and the need to avoid folic acid. This article dives into the science on DHFR genetic variants and how much folic acid is too much.