Key takeaways:
~ Multiple chemical sensitivity is a systemic response to common environmental chemicals that don’t normally cause reactions.
~ Genetic research points to interactions between detoxification genes and the olfactory system.
~ Targeting the right genetic pathways may help alleviate reactions.
What is Multiple Chemical Sensitivity (MCS)?
Multiple chemical sensitivity is a condition in which low-level exposures to common environmental chemicals cause systemic reactions. For example, some people are sensitive to smells such as pesticides or solvents, causing headaches, sinus drainage, joint pain, and more.
Multiple chemical sensitivity (MCS) is also known in research studies as Chemical Sensitivity, Chemical Intolerance, Idiopathic Environmental Illness, and Toxicant-Induced Loss of Tolerance. It is sometimes considered a part of Sensitivity-Related Illnesses or mold reactions.
MCS is often initially caused by a sensitizing event, such as a strong exposure to a specific chemical. It may then shift to include a generalized response that is triggered by a larger set of chemicals.[ref]
Many researchers consider MCS to be a loss of tolerance to environmental contaminants, such as organophosphates, solvents, and heavy metals, even at concentrations considered non-toxic for most people.
Symptoms of Multiple Chemical Sensitivity include:
| Symptom | Description/Example |
|---|---|
| Hypersensitivity | Smell of chemicals, scents seem strong |
| Headaches | Often after exposure |
| Fatigue | More than tiredness, brain fog |
| Respiratory Issues | Chest/throat pain, watery eyes upon exposure |
| Joint Pain | Discomfort continues after exposures |
| Mood/Cognitive | Brain fog, anxiety, depression |
Research on people with MCS shows that it is slightly more common in women than in men.
A recent cohort study looked at the physiological differences between people with MCS who had no other comorbidities (e.g., no fibromyalgia or autoimmune diseases) and an age- and health-matched control group. People with MCS were more likely to have lower blood pressure, lower handgrip strength, and higher BMI.[ref]
Common Triggers and Environmental Factors
Some of the commonly cited offenders include new cars, new furniture, new clothes, mold, carpeting, dust, anything with artificial fragrances, car exhaust, food preservatives, chlorinated water, cigarette smoke, detergents, skin care products, and paint.
Volatile organic compounds (VOCs) are often released indoors from furniture and building products. Research shows VOC levels are ~4 times higher indoors. VOCs often trigger reactions in people with MCS.
Semi-volatile organic compounds (SVOCs) are also released indoors from cleaning products, detergents, synthetic fragrances, pest control sprays, furniture, and plastics.[ref]
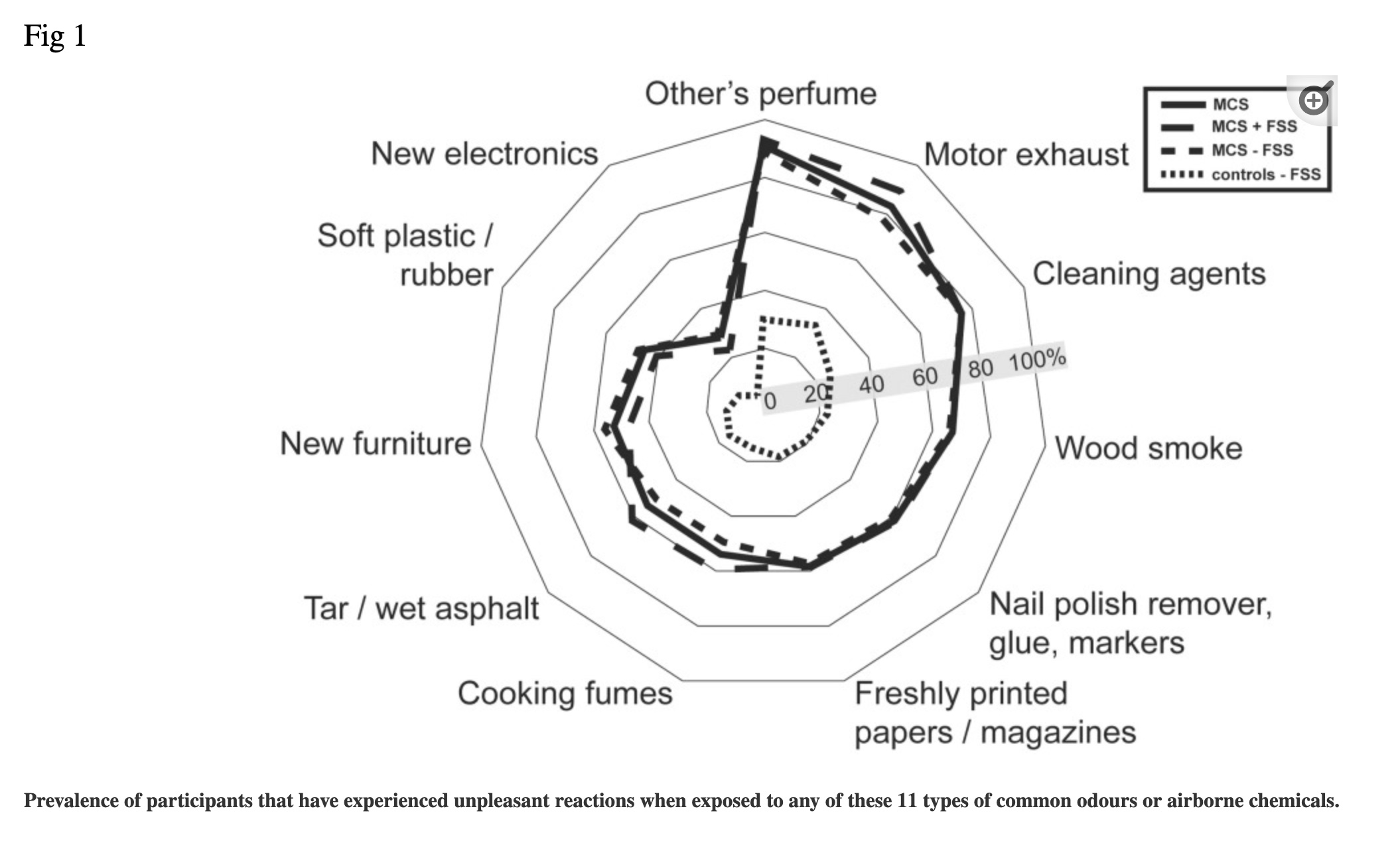
Here is a graph of common triggers from a Danish study on MCS:[ref]
History of MCS:
Multiple Chemical Sensitivity was first described in the 1950s by an allergist named Theron Randolph. Much of what Randolph wrote is considered pseudoscience by the medical community, and the American Medical Association still doesn’t recognize MCS as a disease.
Other countries, such as Germany, Denmark, Finland, Spain, and Austria, do recognize MCS as a disease. The CDC recognizes multiple chemical sensitivity as part of ME/CFS.[ref][ref]
Prevalence:
Studies on MCS are all over the place as to how common it is, which is likely due to different ways of defining the condition. MCS is estimated to affect 0.5-6.5% of the medically diagnosed population, while self-diagnosed cases make up 9.0-11.2% of the general population, according to one paper.
The lack of consensus on diagnostic criteria makes it difficult to know exactly how many people have MCS. A more recent Danish population-based cohort study put the prevalence at 1.95%, while a recent study in the U.S., Australia, UK, and Sweden reported that 7.4% of the population had MCS.[ref]
Is multiple chemical sensitivity psychosomatic?
For many years, the lack of recognition by medical authorities has led many people to question whether MCS is “all in their head” or psychosomatic. Labels such as psychogenic or psychosomatic are applied by doctors, in part, because of a statistical correlation with higher levels of depression and anxiety.[ref]
However, the physiological and cellular changes in people with chemical sensitivities are measurable, statistically significant, and visible, strongly suggesting a physiological cause rather than a psychological cause.[ref][ref]
On the other hand, there are several clinical trials using specific solvents or chemical odors that found no statistical change in measured parameters (e.g. blood pressure, heart rate). [ref][ref] However, the lack of change in autonomic function due to a specific chemical may not be the right variable to control for. People with MCS tend to react systemically in different ways and to different chemicals due to individual past exposures.
Underlying biology in MCS:
Let’s dive into more of the research on what is going on in the body with MCS.
Olfactory system and detoxification:
People with multiple chemical sensitivity are often very sensitive to odors, prompting a lot of research into whether MCS is caused by genetic or other changes to the olfactory system.
Odors are simply the way our brain perceives the stimulus of a specific compound binding to an olfactory receptor in the nose.
We have a number of different odor receptors that detect different compounds. When these receptors are activated in the nose, they send a signal to the brain – letting us know that cookies are baking or that the grass has just been cut. However, the same receptors are found in other parts of the body and can bind to compounds.
Studies of odor detection thresholds don’t show a statistically significant difference in people with MCS. Genetic studies also don’t show that genes related to odor receptors are associated with MCS. Thus, it doesn’t seem likely that hypersensitivity to odors is the problem.[ref]
Instead, much of the research points to hyper-reactivity to certain compounds, possibly due to altered detoxification pathways.[ref]
Oxidative Stress in Multiple Chemical Sensitivity:
Scientists have approached MCS from many angles, using genetic, metabolic, immunologic, etiologic, symptomatic, therapeutic, and epidemiologic tools.
The role of oxidative stress in the development of MCS is now widely accepted, along with alterations in redox status that contribute to chronic mild systemic inflammation.
Patients with MCS exhibit elevated oxidative status and inflammatory markers compared to their healthy counterparts.[ref]
MCS patients also have lower glutathione levels.[ref] Glutathione is a naturally produced antioxidant and one of the main ways that our cells fight oxidative stress.
Genetic variants related to phase I and phase II detoxification of xenobiotic compounds are linked to multiple chemical sensitivity. Phase I and Phase II detoxification are two stages of the body’s natural process for breaking down and eliminating potentially harmful substances, including drugs, toxins, and metabolic waste products.
| Phase | Enzymes | Action | Result |
|---|---|---|---|
| Phase I | CYP450 | Makes toxins more reactive and polar | Prepares for Phase II |
| Phase II | GST, UGTs, SULTs others | Water-soluble conjugates for elimination | Excretes via urine/sweat |
A mismatch between the breakdown rate of compounds in phase I and phase II can cause oxidative stress in the cells. For example, if a phase I metabolite can’t be cleared out quickly enough with phase II conjugation, then it can cause oxidative stress.
One way that this could cause the symptoms seen in MCS is that oxidative stress can cause neurogenic inflammation due to the vulnerability of the central nervous system to free radicals.[ref]
Combination of oxidative stress and odorant reactions?
Another possibility involves the detoxification genes acting on odorants within the nose – or, rather, not responding to odorants appropriately.
Linking the olfactory system and the detoxification system, animal studies show that GST (glutathione s-transferase) and CYP450 enzymes actually participate in odorant clearance and the olfactory process.
The GST genes are expressed in the nasal mucosa. Experiments have shown that odors that are glucuronoconjugated (using the UGT genes) elicit the lowest olfactory response. Other experiments show that the GST enzymes are essential for how animals detect odors and respond to pheromones.[ref]
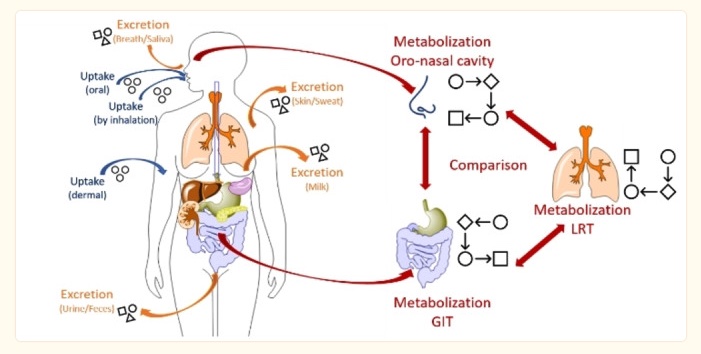
Transdermal route:
Odorants are inhaled, of course, but some, such as essential oils, sulfur compounds, and terpenes, can also be absorbed through the skin. Once an odorant is absorbed into the body, it is either distributed in the circulation or biotransformed (e.g. by CYP 450 enzymes).
Stored in fat:
Interestingly, lipophilic (fat-soluble) odorants are often stored in your fat tissue.[ref]
A recent Danish study found that people with MCS were more likely to have a higher waist circumference and body fat percentage than an age and health-matched control group without MCS.[ref] Perhaps this indicates that more of the lipophilic odorants are stored in fat.
TRPs in MCS: Ion channel activation
Transient receptor potential (TRP) receptors are a family of ion channels expressed in the nervous system. These receptors act as cellular sensors of physical stimuli.
Imaging studies of the brains of people with MCS show that TRPV1 and TRPA1 are overly sensitized.[ref]
- TRPV1 senses volatile compounds such as toluene, styrene, benzene, ethylbenzene, acetone, diethyl ether, hexane, heptane, cyclohexane, and formaldehyde. TRPV1 is also activated by capsaicin (hot peppers), low extracellular pH, and noxious heat.[ref]
- TRPA1 is activated by VOCs, cigarette smoke, heavy metals, naphthalene (moth balls), formaldehyde, and styrene.[ref]
TRP receptors are highly expressed in the olfactory and trigeminal nerve endings and in the brain. TRPV1 is expressed in about 60% of vagal neurons, and TRPA1 in about 25%.[ref] In addition to chemical stimuli, TRP receptors respond to oxidative stress and systemic inflammation.[ref]
Repeated activation and hyperexcitability:
TRPA1 and TRPV1 receptors are often co-localized on the same cells. Repeated activation of the receptors can lead to upregulation and sensitization, causing hyperexcitability of the receptor even at low levels.
Importantly for MCS, the two receptors modulate and sensitize each other. For example, the sensitization of TRPA1 with a low dose exposure to acrolein causes sensitization of TRPV1 against capsaicin.[ref]
Studies in workplaces that have high VOC levels show that TRPA1 activation is “the molecular initial event (MIE) leading to sensory irritation.”[ref]
A 2022 study investigated the sensitivity to capsaicin, a TRPV1 receptor activator, in people with MCS compared to a control group. The results showed that capsaicin provoked significantly more symptoms in MCS patients than in healthy controls.[ref]
Mold, infections, and pesticides:
Mold toxins also trigger MCS in some people. Exposure to “dampness microbiota” in a building that has moisture problems increases the relative risk of multiple chemical sensitivity by about 3-fold.[ref]
Research also shows that having a significant infection in the past decade, either viral or bacterial, is linked to an increased relative risk of MCS. [ref]
One study of people who react to building interiors looked specifically at acrolein, an aldehyde that has an acrid odor. Acrolein is used as a herbicide and algaecide, but it is also produced by the burning of fossil fuels, cigarette smoke, burning fats, and the oxidation of volatile organic compounds that are emitted in new construction. The people in the study with known chemical sensitivities detected acrolein at low levels and had more eye, nose, and throat symptoms after about 30 minutes of exposure.[ref]
Gut microbiome and MCS:
A study involving 30 adults with MCS in Japan looked at the gut microbiome composition and compared it to an age and sex-matched control group. The results showed that “Akkermansia muciniphila was significantly more abundant (p = 0.02, fold change = 3.3) and Faecalibacterium prausnitzii significantly less abundant in MCS patients than in HC (p = 0.03, fold change = 0.53).” They also found that xylene and dioxin degradation pathways were upregulated.[ref]
Interestingly, Akkermansia muciniphila is currently being promoted as a probiotic. It is associated with healthy body weight, and it produces short-chain fatty acids that are used by other gut microbes. However, Akkermansia muciniphila also eats your gut mucosal layer. It could be that the 3-fold higher abundance of Akkermansia muciniphila in MCS patients decreases their gut mucosal barrier and activates the body’s immune response.[ref]
Now let’s take a look at how your genetic variants increase or decrease your susceptibility to MCS.
Multiple Chemical Sensitivity Genotype Report:
Access this content:
An active subscription is required to access this content.
Lifehacks: Natural Solutions for Multiple Chemical Sensitivity
Lifestyle changes:
If you have been dealing with MCS for a while, you probably already know and understand which chemicals are likely to trigger symptoms. However, here are some suggestions that may help if you are new to MCS. Of course, talk to your doctor for any medical advice.
Improve indoor air quality:
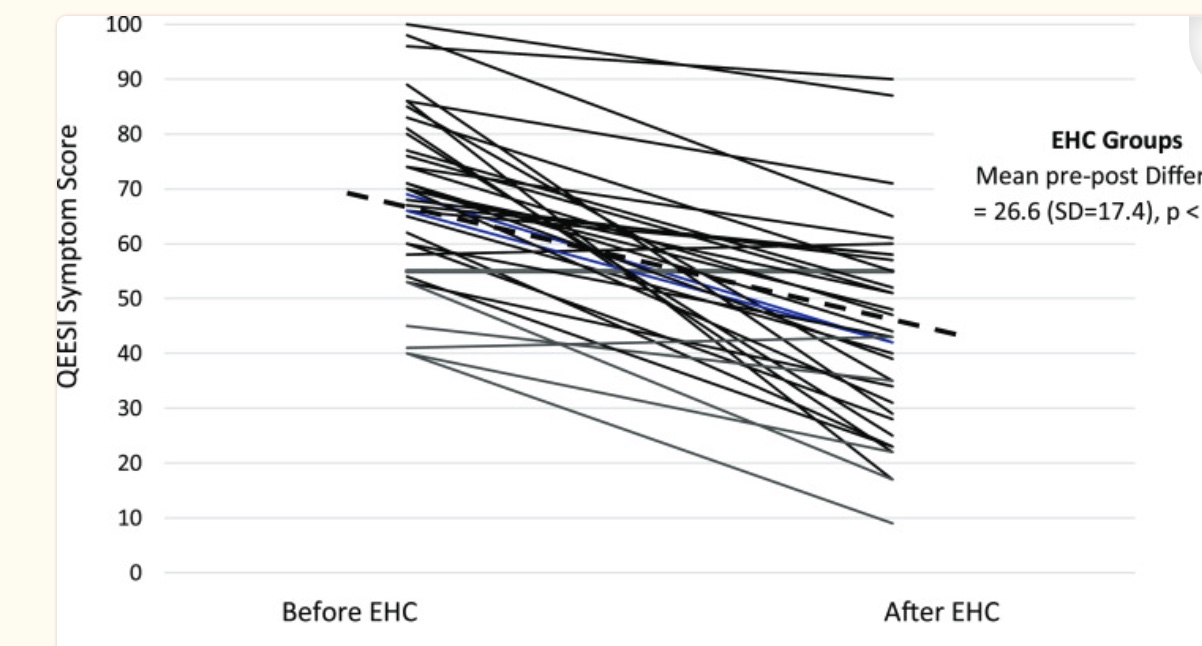
Studies show that improving indoor air quality can help significantly reduce MCS symptoms. The “Environmental House Call” study looked at the effects of recommended interventions such as eliminating cleaning products, personal care products, and laundry products with fragrances and VOCs. Additionally, carpets and major outgassing sources were removed. [ref]
Laundry detergents and fabric softeners:
Laundry products are notorious for fragrances that trigger MCS symptoms. Look for detergents that say “Free and Clear” or “Free and Clear with Odor Remover”. Keep in mind that fragrance-free cleaners and laundry products may still contain VOCs.[ref]
- Try washing with vinegar, baking soda, or soap nuts (if you are comfortable with the natural scent).
- Keep in mind that it may take several washes to remove previously scented detergent and fabric softener odors from clothing.
Home and travel:
There’s a website called Find a Pure Hotel Room that lists hotels that are free of fragrances and contaminants. This may be a good place to check if you are planning to travel.
My Chemical-free House is a website run by someone with MCS. It has many suggestions for non-toxic and scent-free home and building products.
Air purifiers:
Reducing indoor air pollution from off-gassing and VOCs can be helpful, and air purifiers are a good option here. HEPA filters and UV light can help if mold spores are one of your triggers.
Organic:
Eating organic whenever possible helps to reduce exposure to pesticide residues on food.
Natural fragrances:
Essential oils and natural fragrances may still bother some people with MCS, so look for personal care products that are labeled fragrance-free rather than with natural scents.
Genetic connections:
Let’s look at possible solutions for specific genetic variants.
GSTM1 null variant:
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
References: