Key takeaways:
~ Progesterone is an important hormone in women’s health, regulating menstruation, and for pregnancy.
~ Progesterone is important for everyone, men and women, for brain health and overall well-being.
~ Genetic variants play a role in how your body makes progesterone, how it uses progesterone, and how it breaks down and eliminates progesterone.
What does progesterone do in the body?
Progesterone is a hormone made by both men and women. It plays a greater role in premenopausal women, and while that will be the main focus here, the same mechanisms take place for men to a lesser degree.
As a hormone, progesterone can interact with different receptors in the nucleus of cells and turn on or off the production of other genes. Progesterone can also act directly on non-nuclear receptors in the ovaries.[ref]
In the brain, progesterone acts on the HPA axis (hypothalamus, pituitary, adrenals) and regulates the production of luteinizing hormone (LH) in women. This is a feedback loop between the ovarian production of progesterone and the control of LH from the hypothalamus.
Progesterone also affects mood and the way people feel and behave. In general, progesterone reduces sexual desire in women as well as decreases aggression and anxiety. When it comes to PMS (irritability, anxiety, and aggression), there is a lower-than-normal level of progesterone.
In the brain, progesterone metabolizes into allopregnanolone, which is neuroprotective in brain trauma.[ref]
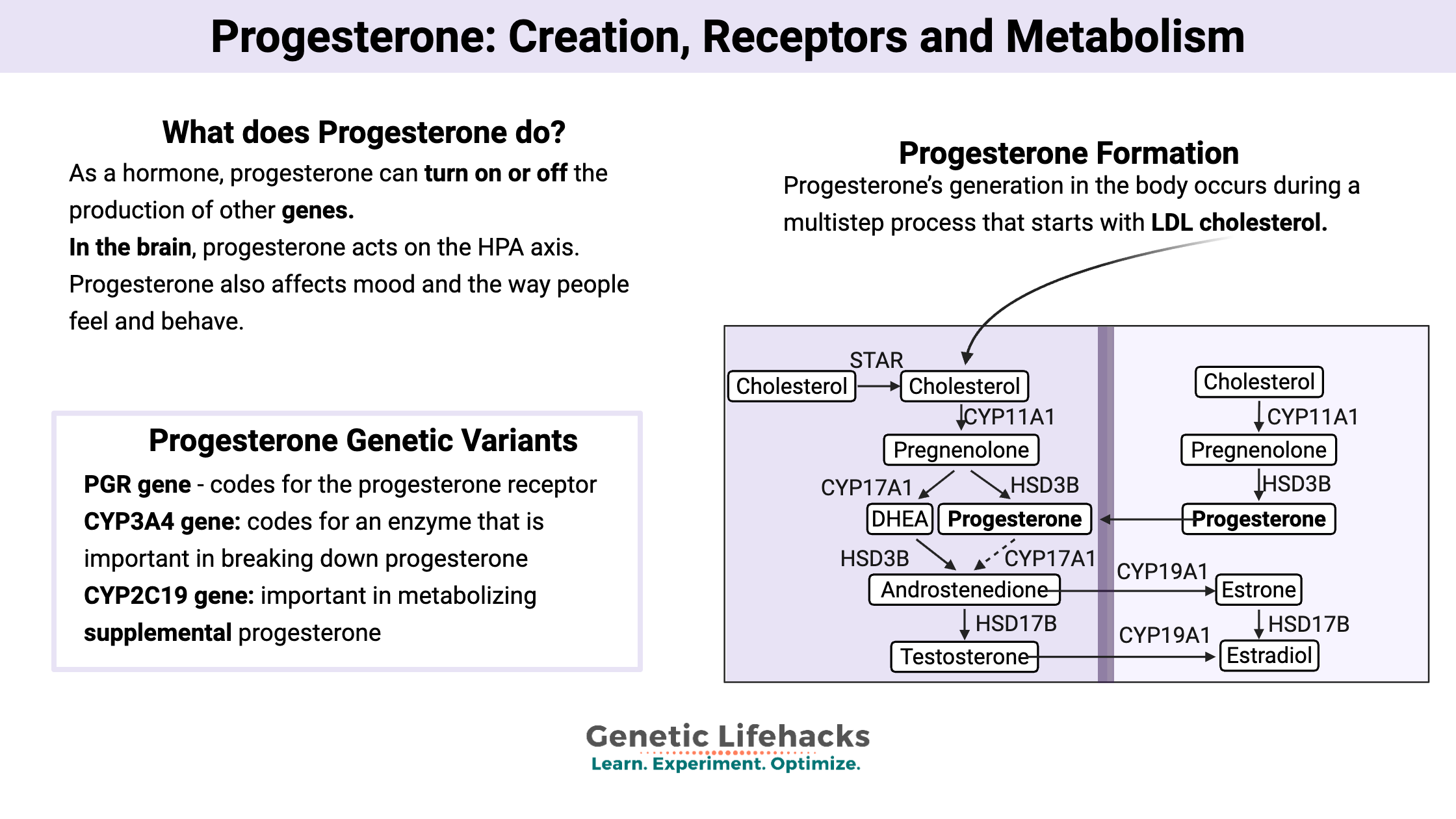
How is progesterone made?
Progesterone’s generation in the body occurs during a multistep process that starts with LDL cholesterol.
The LDL cholesterol is taken into cells via cholesterol receptors, where it is transferred into the mitochondria to be converted into pregnenolone.
Pregnenolone is released by the mitochondria and can take two paths, depending on the organ where it is created:[ref]
- In the adrenals, pregnenolone turns into DHEA, androstenediol, and 17-alpha hydroxypregnenolone. From here, it generally gets converted into glucocorticoids and androgens.
- In the female reproductive organs, pregnenolone becomes progesterone and 17-alpha hydroxypregnenolone. From here, it can become androstenedione and then estrogens. Progesterone from the reproductive system can circulate in the bloodstream, where it can then act on the regions of the brain that are controlling the reproductive hormones.
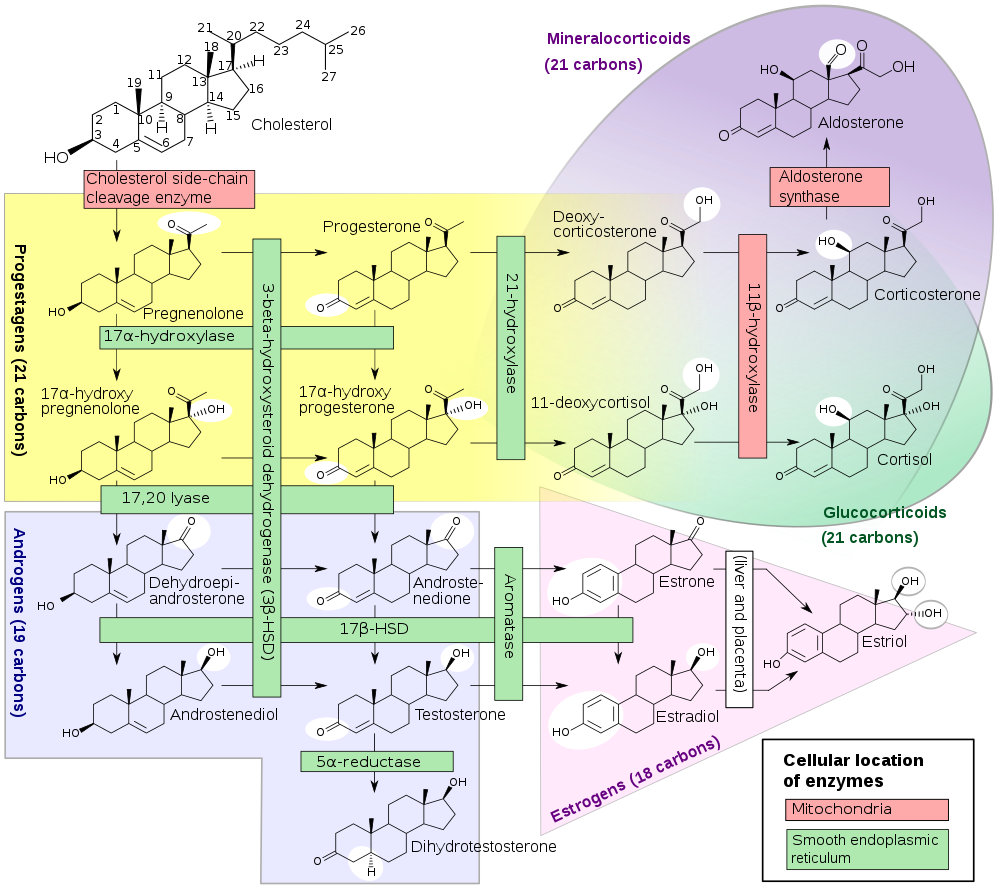
Diagram of steroid hormone production[ref]:
Mechanism of action for progesterone:
Let’s get into the details of how progesterone works to turn on and off genes.
Progesterone is a lipophilic hormone, so it can diffuse across cell membranes. Within the nucleus of the cell, it attaches to progesterone receptors. These progesterone receptors come in a couple of different sizes and are denoted as PR-A and PR-B. The different flavors of receptors are active in different tissues. PR-A is important for ovulation in the ovaries, and PR-B is part of the development of mammary glands.[ref]
Progesterone Receptor Types
| Receptor | Location/Function |
|---|---|
| PR-A | Ovary (ovulation) |
| PR-B | Mammary glands (development) |
| Membrane-associated | Cholesterol/steroid metabolism, iron balance |
Once progesterone binds to the receptor, it will bind to certain spots on the DNA to recruit other proteins to the site. These regulatory proteins then activate or repress the transcription of certain genes.
It gets a little complex – but just think of it as progesterone causing other genes to get turned on (via the progesterone receptor). These other genes help prepare the uterus for pregnancy, like growth factors needed for regenerating the uterine lining.[ref]
Interestingly, other genes regulated by progesterone include a couple of important circadian rhythm genes.
Additionally, progesterone can act on specific membrane-associated progesterone receptors. These receptors affect cholesterol and steroid biosynthesis, metabolism, as well as balancing iron.[ref]
Balancing act: Estrogen vs. Progesterone
Progesterone plays a role in preparing the uterus for implantation of the fertilized egg, and then, if pregnancy occurs, progesterone levels rise over the course of the pregnancy and are essential for maintaining the pregnancy.[ref]
In a sense, progesterone is balancing out some of the growth that estrogen is promoting. Estrogen stimulates the endometrium, and progesterone is coming along to regulate the gene expression of the fibroblast growth factors.[ref]
Table: Progesterone vs. Estrogen
| Estrogen | Progesterone |
|---|---|
| Stimulates endometrial growth | Regulates endometrial growth, prepares uterus for implantation |
| Promotes fibroblast growth factors | Regulates gene expression of growth factors |
| Increases progesterone receptor expression | Inhibits estrogen receptor expression |
When not pregnant, menstruation occurs when progesterone levels drop at the end of the menstrual cycle. The black line in the image below shows how progesterone rises after ovulation (by 10 to 35-fold) and drops at the start of menstruation.[ref]
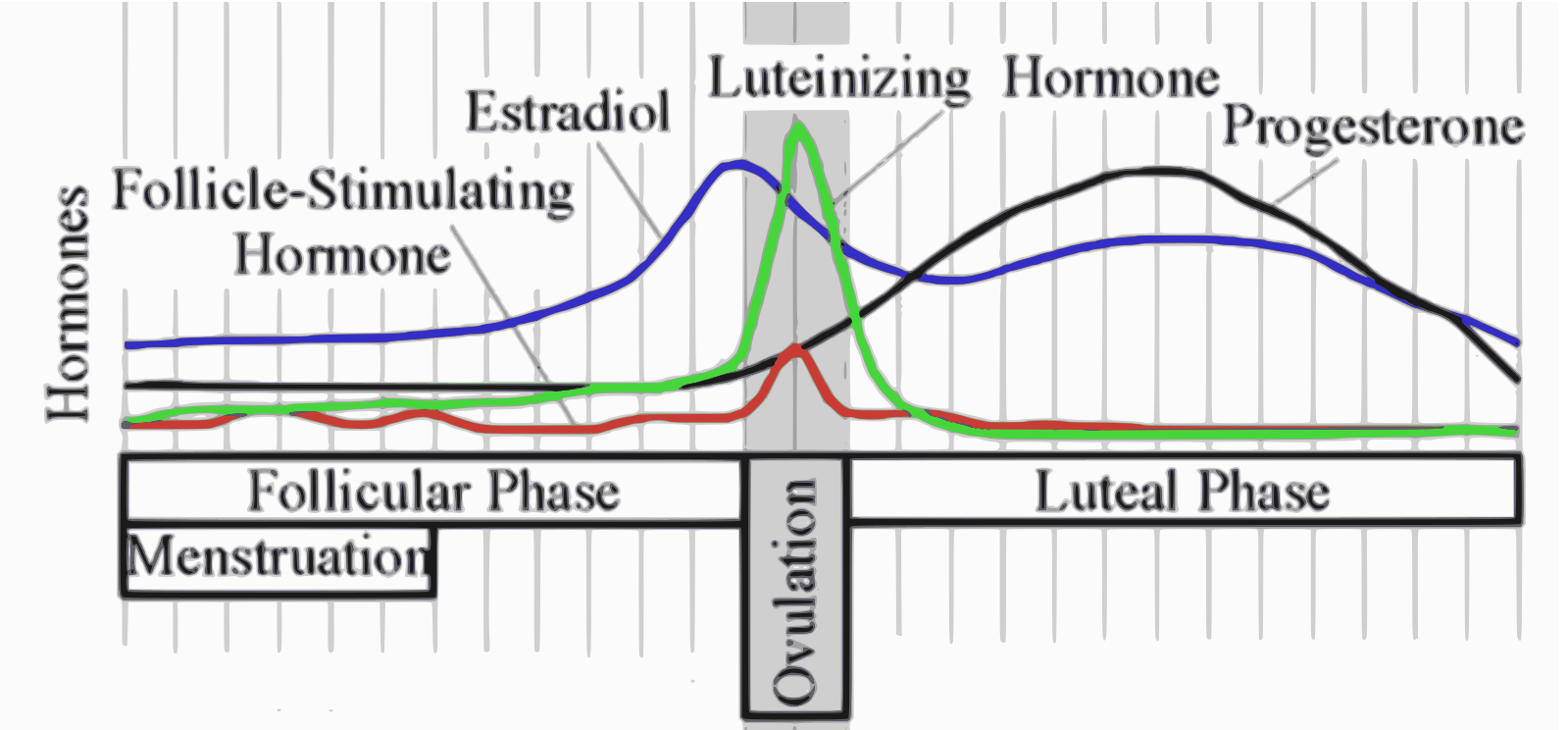
In the uterus, progesterone receptor expression is dependent on and responsive to the amount of estrogen. Additionally, estrogen receptor expression is inhibited by progesterone, forming a feedback loop.[ref]
Metabolism of progesterone:
Like all hormones created in the body, progesterone needs to be broken down (metabolized) and eventually excreted or recycled.
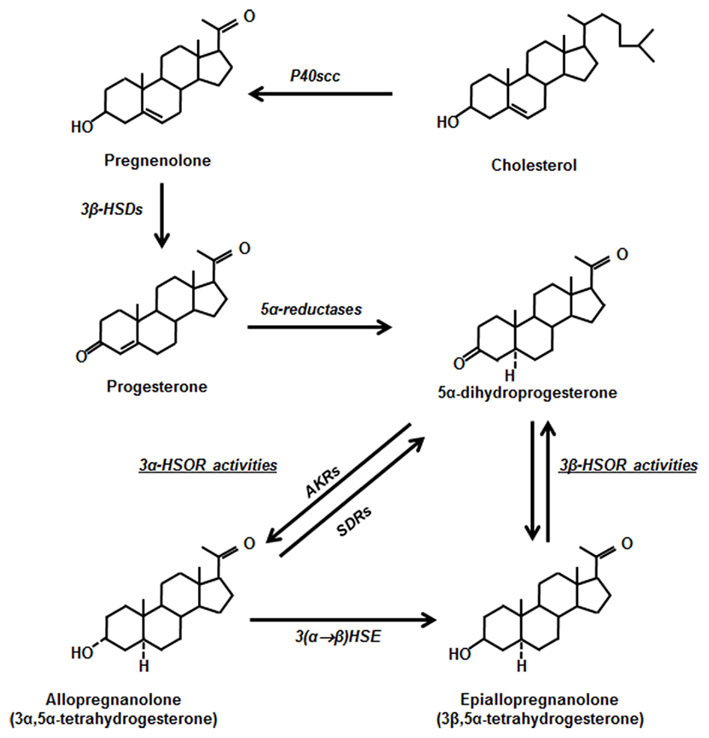
The metabolites of progesterone can also play an active role in the body. For example, allopregnanolone is important in brain health.
The steroid enzyme 5α-reductase (5αR) acts on progesterone, converting it to 5α-dihydroprogesterone (DHP). 5αR also metabolizes testosterone and deoxycorticosterone.
DHP, the progesterone metabolite, is active in the brain as a neuroactive steroid that modulates the GABA-A receptors. (Read all about GABA here) Both progesterone and the DHP metabolite are important in the brain. Animal studies show that changing 5αR levels (via drugs such as finasteride) affects behavior and brain function.[ref]
Additionally, progesterone and synthetic progestin are metabolized through the CYP450 enzymes, a family of enzymes responsible for breaking down many substances made by the body as well as toxins and medications. CYP3A4 is the main route of metabolism[ref], and CYP2C19 also plays a role.
~ Metabolized by 5α-reductase and CYP450 enzymes (mainly CYP3A4, CYP2C19)
~ Produces neuroactive metabolites like DHP and allopregnanolone
Progesterone pills and supplements:
Progesterone can be a part of birth control pills (estrogen + progesterone), or it can be given alone (e.g., Depo-Provera birth control shots).
There are four main ways of getting additional progesterone (synthetic progestins or bioidentical) into the body:[ref]
- Transdermal progesterone cream is generally not enough to raise the plasma concentration of progesterone significantly – at least not compared with other methods.
- The absorption of oral progesterone pills occurs in the intestines. It then travels to the liver and where it mostly breaks down. Thus, oral doses of progesterone must be really high to get any of it past the liver.
- Vaginal progesterone suppositories make it into the uterus and endometrium more readily.
- Progesterone shots work to increase progesterone levels, but there is poor compliance.
Normally, progesterone rises to high levels during pregnancy, which is essential for maintaining pregnancy. Sometimes women with a high risk of miscarriage are given progesterone.
Blocking the progesterone receptor is a way to terminate a pregnancy during the first two months. Mifepristone, or RU-486, is the name of this drug.
Ella (ulipristal acetate) is another medication that acts on the progesterone receptor.[ref] Known as the morning-after pill, Ella is sold as a prescription emergency contraceptive in the US.
Synthetic progesterone medications known as progestins or progestogens are often used as birth control or for menopausal hormone therapy.
Etonogestrel, a synthetic progestin, can be implanted into the arm for 1-3 years of birth control. Some people carry an extra copy of the CYP3A7 gene. This gene codes for an enzyme needed for a baby’s development but usually switches off at birth. About 4% of the population carries an additional copy of the CYP3A7 gene (not covered by AncestryDNA or 23andMe data), which affects the metabolism of progestins (progestogens) as well as other steroid hormones. It can also affect the efficacy of etonogestrel as a birth control method.[ref]
Table: Progesterone Supplementation Methods
| Method | Notes/Effectiveness |
|---|---|
| Transdermal cream | Generally insufficient to significantly raise blood levels |
| Oral pills | High first-pass metabolism in liver, requires high doses |
| Vaginal suppositories | Direct effect on uterus/endometrium |
| Injections (shots) | Effective, but compliance may be poor |
Progesterone Genotype Report:
Access this content:
An active subscription is required to access this content.
Lifehacks:
Please be sure to talk with your doctor if you have questions on whether supplemental progesterone is right for you. Your doctor can also help you with testing your progesterone and estrogen levels. Below are some studies that shed light on the topic.
Studies on supplemental progesterone:
Bioidentical progesterone creams:
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
References: