Key takeaways:
- TNF-alpha (tumor necrosis factor alpha) is an inflammatory cytokine produced by the immune system to fight off pathogens and cancer cells.
- Chronically elevated TNF-α levels are linked to autoimmune, cardiovascular, neurodegenerative disease, gum disease, and mood disorders.
- Genetic variants can increase your susceptibility to chronically elevated TNF-alpha.
- Understanding your TNF gene variants can help you find targeted, natural solutions for chronic inflammation.
What is TNF Alpha?
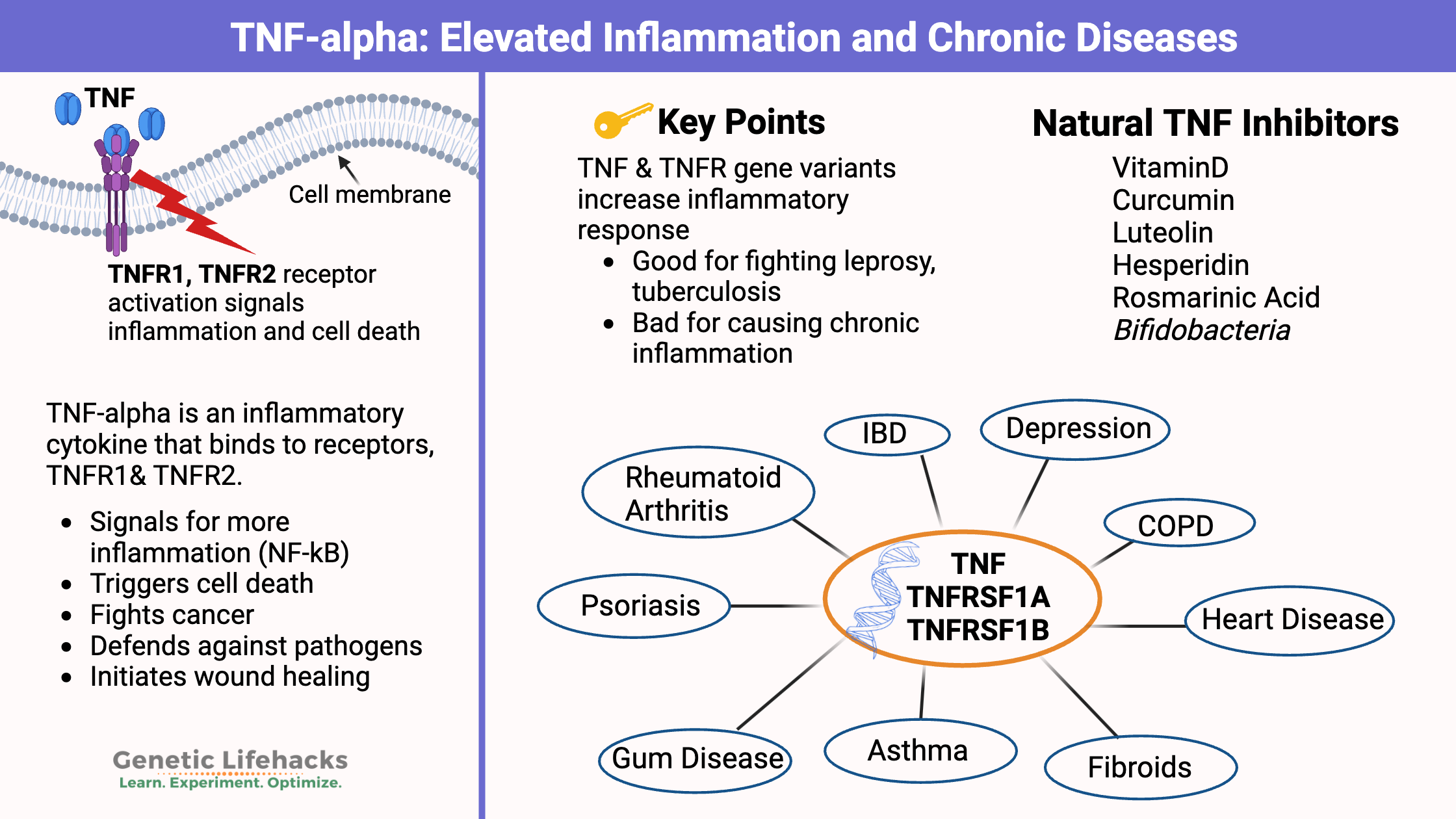
TNF-alpha is an inflammatory signaling protein that your immune system uses to fight infections, heal wounds, and help destroy cancer cells. When TNF-alpha stays too high for too long, it can drive chronic inflammation linked to autoimmune disease, heart disease, mood disorders, and neurodegenerative conditions. Common TNF gene variants can make some people produce more TNF-alpha than others, which may increase their risk for these chronic diseases.[ref]
Let’s go through some background science on what TNF-alpha does, how it increases inflammation, and why it can be a significant problem. Then we will look at the genetic variants that increase TNF levels and the diet, lifestyle changes, and supplements to prevent high TNF.
Inflammatory signaling molecule: TNF calls up the troops
We think of redness, swelling, heat, and pain as inflammation, such as what happens after getting injured. However, the inflammatory response is also vital in fighting off bad bacteria, viruses, or fungi.
When the body needs an inflammatory response to fight off an invader, it releases inflammatory cytokines, such as TNF-α (and others).
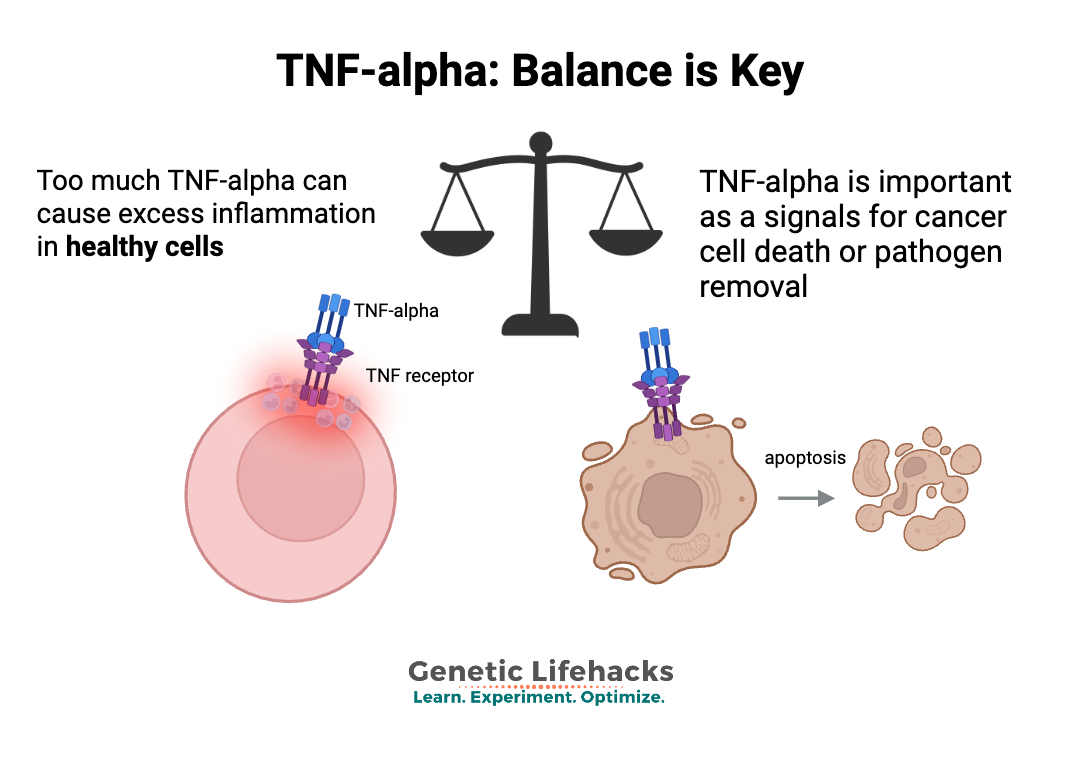
When TNF-alpha binds to its receptor on the surface of a cell, two actions can occur:
- One option is that it kills the cell (apoptosis). It’s like it pulls the pin on a grenade.
- Another option is that it can also cause the cell to produce other inflammatory response molecules, but the cell still survives.
Cancer killing:
While cell death sounds bad, it is completely necessary to fight off certain infections — or if a cell is cancerous. Thus, the name tumor necrosis factor (TNF) indicates that it kills tumor cells.
Wound healing:
TNF-α is also essential for the initial stages of wound healing. Inflammation is a necessary step for healing injured tissue.[ref]
Pathogen defense:
TNF-alpha is an essential part of the body’s initial defense against pathogens such as:[ref][ref][ref][ref]
- Tuberculosis
- Histoplasmosis
- Malaria
- Leprosy
TNF-α: Signaling Molecule that Binds the TNF Receptor
TNF-α is synthesized and released by activated immune cells – macrophages, lymphocytes, mast cells, and neutrophils. It is also produced as a transmembrane protein, as well as by smooth muscle cells in response to injury.[ref][ref]
After being released by a cell (e.g. mast cell or a macrophage), TNF-α can bind to several different receptors on the surfaces of cells.
The two main TNF-alpha receptors are TNFR1 and TNFR2. Most cells in the body have the TNFR1 receptor, but the TNFR2 receptor is more specific to immune system cells.[ref] Genetic variants in the TNF receptors can increase or decrease your risk of inflammatory conditions. (see the genotype report section below for your variants)
When TNF binds to a receptor, several different actions can occur, depending on the cell type and receptor type:
- activates NF-κB, which is a transcription factor that controls cell survival and inflammatory response
- activates the MAPK pathways, which are important in the cell cycle and in preventing cancer
- signals for cell death
The TNF receptors are a hot topic of research for therapeutics to treat autoimmune diseases. Currently, researchers are looking at monoclonal antibodies, peptides, and small molecules that can change the TNF receptor in a way that TNF-alpha can’t bind to it.[ref]
How does TNF-alpha cause inflammation?
The immune response is all about balance. You want to fight off pathogens with a strong response, but you don’t want a constant inflammatory response continuing when it isn’t needed.
Super fighter:
Genetic variants that increase TNF-alpha levels are linked to being better able to fight off pathogens, such as malaria or hepatitis B.[ref]
But.. That superpower of fighting pathogens and killing cancer cells comes with a price. For example, chronically elevated levels of TNF-alpha are linked with an increased risk of autoimmune diseases, skin infections, and gum disease.
Chronic inflammation in the body: Tradeoffs with pathogen defense
Several common genetic variants increase TNF-alpha levels and increase the risk of inflammatory conditions. Keep in mind that these same genetic variants that helped your ancestors survive leprosy or tuberculosis (and thus lived to pass on the variant to you), could be at the root of many of the inflammatory conditions that plague us today.[ref]
Conditions linked to chronically elevated TNF:
Elevated TNF-alpha is linked to the following chronic conditions:
| Disease/Condition | Role of High TNF-alpha | Key Mechanism/Notes |
|---|---|---|
| Rheumatoid Arthritis (RA) | Drives joint inflammation, tissue destruction | Autoimmune pathway, synovial swelling |
| Inflammatory Bowel Disease (IBD) | Promotes intestinal inflammation, ulceration | Chronic immune activation in gut |
| Lupus (SLE) | Increases systemic inflammation, organ damage | Autoimmune, multi-system involvement |
| Psoriasis | Stimulates keratinocyte proliferation, skin plaques | Chronic skin inflammation |
| Multiple Sclerosis (MS) | Contributes to demyelination, neuronal loss | CNS neuroinflammation |
| Alzheimer’s Disease | Aggravates neuroinflammation, promotes amyloid pathology | Neuronal cell death, cognitive decline |
| Parkinson’s Disease | Increases neuroinflammation, dopaminergic neuron loss | CNS cell death, motor impairment |
| Depression/Mood Disorders | Dysregulates HPA axis, cortisol, neurotransmitters | Links mood, inflammation, and stress |
| Cardiovascular Disease | Accelerates atherosclerosis, vascular inflammation | Promotes plaque formation and rupture |
| Non-alcoholic Fatty Liver Disease | Induces hepatic inflammation, fibrosis | Steatosis, risk for cirrhosis |
| Gum Disease/Periodontitis | Increases inflammatory response in gums | Tissue destruction, tooth loss |
| Skin Infections | Exaggerates local inflammatory response | Chronic wounds, poor healing |
| Obesity-related Metabolic Syndrome | Sustains adipose inflammation, insulin resistance | Links TNF-alpha with metabolic changes |
Let’s take a look at some of the studies and mechanisms of action involved with chronically elevated TNF.
TNF-alpha and brain health:
Blood-brain barrier (BBB) and neuroinflammation: Elevated TNF-alpha in the body increases the permeability of the BBB. This leakiness of the blood-brain barrier then allows inflammatory mediators to cross into the brain and activate more inflammation in the brain. In the brain, TNF-alpha causes elevated reactive oxygen species and impaired insulin signaling, leading to more neuroinflammation.[ref] This can be a cause of brain fog, for some people.
Related article: Brain fog genes
Inflammation and Depression: Recent research shows that, for some people, pro-inflammatory cytokines are at the heart of a major depressive disorder. For a subset of patients with depression, TNF-alpha is elevated, and blocking TNF-α can ameliorate depressive symptoms. A 2008 genome-wide association study found that a TNF genetic variant increases the risk of depression.[ref] A recent study in patients with rheumatoid arthritis showed that a positive side effect of TNF inhibitor drugs was a decrease in depression.[ref]
Related article: Depression and Inflammation
HPA axis and cortisol: Researchers think that inflammation, specifically high TNF alpha levels, impacts the HPA axis and elevates cortisol. HPA axis dysfunction is strongly linked to depression. Additionally, higher TNF-alpha levels can cause increased uptake of serotonin into cells, causing depressive symptoms.[ref]
Related article: HPA axis dysfunction and your genes
Neurodegenerative diseases: Alzheimer’s and Parkinson’s diseases are both linked to higher TNF-alpha production in the brain. Research shows that microglia and astrocytes release TNF-alpha when activated, and this increases the production of amyloid-beta plaque, which is linked with Alzheimer’s disease. Additionally, amyloid-beta can activate microglia and astrocytes, further perpetuating the creation of TNF-alpha and other inflammatory cytokines.[ref][ref]
Related article: Alzheimer’s and APOE genotype
TNF-alpha and chronic conditions:
Heart disease studies: TNF-α is increased in heart disease, raising the question of directionality: whether heart disease causes an increase in TNF-α or whether higher TNF-α contributes to causing heart disease. To answer this question, researchers turned to genetics.
Studies show variants that increase TNF are causally linked to an increased risk of heart disease. Thus, limiting TNF-alpha may help to reduce the risk of heart disease.[ref]
Related article: Genetics and Coronary Artery Disease
Chronic Fatigue: Genetic studies also show that variants in the TNF gene, which encodes TNF-alpha, increase susceptibility to ME/CFS. These variants are associated with increased TNF response to pathogens or mycotoxins.[ref]
Related article: ME/CFS and Genetics: Chronic Fatigue Syndrome Genes, Immune Pathways, and Natural Solutions
Intestinal absorption is altered with high TNF-alpha:
Inflammation and high TNF-alpha levels in the intestines alter the ability to absorb nutrients.
For example:
- High TNF-alpha levels decrease the receptor needed for the absorption of vitamin C (SLC23A1).[ref]
- Short-chain fatty acids, such as butyrate, are important for colon health. High levels of TNF-alpha decrease the expression of transporters for butyrate (SLC5A8).[ref]
- Phosphate absorption is decreased when TNF-alpha is high in the intestines. Low phosphate absorption can cause problems with teeth and bones.[ref]
TNF and metabolic health:
Fatty liver disease:
Non-alcoholic fatty liver disease (NAFLD, MAFLD) is caused by increased fat stored in the liver. This can eventually lead to liver disease. TNF-alpha, as well as a couple of other inflammatory cytokines, are increased in fatty liver disease. Research using animals that were genetically altered to reduce TNF-alpha levels shows that TNF-alpha is a driving factor in fatty liver disease.[ref][ref]
Related article: Fatty liver disease (NAFLD, MAFLD) genes
Uterine Fibroids: Hormones plus TNF-alpha
Uterine fibroids are benign tumors in the uterus. These are a common occurrence, with estimates ranging from 50-80% of women having fibroids at some point in their lives. The growth of the fibroids is thought to be caused by steroid hormones (estrogen, progesterone), growth factors, cytokines, and chemokines. Research points to TNF-alpha as the most important cytokine involved in fibroid growth.[ref]
Related article: Fibroids genes
TNF inhibitors and cancer risk:
TNF-alpha inhibitors are monoclonal antibodies or TNF-i drugs that are used primarily for autoimmune diseases. These types of drugs can cause a degree of immunosuppression in an effort to prevent the symptoms and damage from the autoimmune condition.
TNF – tumor necrosis factor – is also important for killing cancerous cells and preventing cancer, so a natural question is whether TNF inhibitors increase the risk of cancer.
A 2025 study compared the cancer risk for ~13,000 patients on TNF inhibitors with ~37,000 controls who were not on TNF medications. People taking TNF inhibitors were at an increased relative risk of skin cancers, including melanoma and squamous cell carcinoma. On the positive side, those on TNF inhibitors were at a decreased risk of prostate cancer, leukemia, and non-Hodgkin lymphoma. [ref]
TNF Genotype Report
Genetic variants (SNPs) associated with increased TNF-alpha levels:
Here are several genetic variants linked to naturally more active TNF-alpha, which then increases the risk of multiple chronic conditions. On the plus side, higher TNF may help to protect against pathogens.
Access this content:
An active subscription is required to access this content.
Lifehacks: Natural ways to inhibit TNF alpha
If you carry genetic variants related to higher TNF-alpha levels and have a related inflammatory condition, inhibiting TNF-alpha may help to reduce chronic inflammation.[ref]
Keep in mind the tradeoff between TNF-alpha as a response to pathogens and inhibiting TNF-alpha to reduce chronic inflammation. If you are at a higher risk for infections, talk to your doctor.
Lifestyle and diet changes to decrease excess TNF-alpha:
Vitamin D may help people with MS and the TNFRSF1A variant. A study found that there was a statistically significant interaction with variant carriers being at high risk for relapse with low vitamin D levels.[ref] Sun exposure on your skin in the middle of the day is needed for vitamin D production. Lacking that, vitamin D supplements are also available.
Moderate exercise exerts part of its anti-inflammatory effects by reducing TNF-alpha.[ref]
Cigarette smoking is linked to significantly higher TNF-alpha levels, and this association may be a driving factor in COPD and other chronic inflammatory diseases linked to smoking.[ref][ref]
High folate vegetable intake decreased TNF-alpha levels in women with the MTHFR C677T AA genotype.[ref] Check your MTHFR genotype here.
Omega 3 fatty acids (DHA and EPA) from seafood or fish oil help to resolve inflammation and reduce TNF-alpha levels.[ref]
Related article: SPMs and the resolution of inflammation
Microplastics cause TNF-alpha release:
An ever-growing amount of research shows that microplastics and nanoplastics are absorbed into the body and cause inflammatory effects.
- A study on polystyrene microplastics shows that they activate toll-like receptors (TLR4 and TLR2), which then increases TNF-α.
- Studies using vascular smooth muscle cells show that they release TNF-alpha and IL-6 when cultured with microplastics.[ref][ref]
Related article: Microplastics research
10 Natural tumor necrosis factor inhibitors:
Along with the lifestyle changes, certain polyphenol supplements can significantly reduce TNF levels.
Always check with your doctor or pharmacist if you are on medications before beginning a new supplement. Several of these TNF-alpha inhibitors can lower blood pressure and may interact with medications.
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
Frequently Asked Questions about TNF alpha:
What is TNF-alpha?
TNF-alpha (tumor necrosis factor alpha) is an inflammatory cytokine made by immune cells that helps fight infections and cancer cells by triggering inflammation and, in some cases, cell death.
Why is high TNF-alpha a problem?
When TNF-alpha stays chronically elevated, it is linked to autoimmune diseases, cardiovascular disease, depression, neurodegenerative disorders, skin and gum disease, and obesity-related metabolic problems.
Which conditions are associated with elevated TNF-alpha?
Rheumatoid arthritis, IBD, lupus, psoriasis, multiple sclerosis, Alzheimer’s disease, Parkinson’s disease, depression, heart disease, fatty liver disease, gum disease, and metabolic syndrome are all associated with higher TNF-alpha.
Can TNF-alpha affect nutrient absorption?
Yes, high TNF-alpha in the intestines can reduce absorption of vitamin C, butyrate, and phosphate by downregulating their transporters, which may impact gut, bone, and metabolic health.
Related article: Vitamin C: Nutrigenomics, Transport, and Genetic Deficiency
Are TNF-alpha levels influenced by genetics?
Common genetic variants in the TNF gene promoter can increase TNF-alpha production, which may raise susceptibility to autoimmune conditions, infections, and chronic inflammatory diseases.
Can natural compounds help lower TNF-alpha?
Certain natural compounds and lifestyle strategies may modulate TNF-alpha signaling, but they must be balanced against the cytokine’s important role in fighting infections; medical guidance is essential.
References:
Allendoerfer, Ruth, and George S. Deepe. “Blockade of Endogenous TNF-α Exacerbates Primary and Secondary Pulmonary Histoplasmosis by Differential Mechanisms.” The Journal of Immunology, vol. 160, no. 12, June 1998, pp. 6072–82. www.jimmunol.org, https://www.jimmunol.org/content/160/12/6072.
Borthakur, Alip, et al. “The Probiotic Lactobacillus Plantarum Counteracts TNF-{alpha}-Induced Downregulation of SMCT1 Expression and Function.” American Journal of Physiology. Gastrointestinal and Liver Physiology, vol. 299, no. 4, Oct. 2010, pp. G928-934. PubMed, https://doi.org/10.1152/ajpgi.00279.2010.
Chen, Huacong, et al. “Tumor Necrosis Factor-Alpha Impairs Intestinal Phosphate Absorption in Colitis.” American Journal of Physiology. Gastrointestinal and Liver Physiology, vol. 296, no. 4, Apr. 2009, pp. G775-781. PubMed, https://doi.org/10.1152/ajpgi.90722.2008.
Chen, Yihe, and Kuifen Ma. “NLRC4 Inflammasome Activation Regulated by TNF-α Promotes Inflammatory Responses in Nonalcoholic Fatty Liver Disease.” Biochemical and Biophysical Research Communications, vol. 511, no. 3, Apr. 2019, pp. 524–30. PubMed, https://doi.org/10.1016/j.bbrc.2019.02.099.
Ciebiera, Michał, et al. “The Role of Tumor Necrosis Factor α in the Biology of Uterine Fibroids and the Related Symptoms.” International Journal of Molecular Sciences, vol. 19, no. 12, Dec. 2018, p. 3869. PubMed Central, https://doi.org/10.3390/ijms19123869.
—. “The Role of Tumor Necrosis Factor α in the Biology of Uterine Fibroids and the Related Symptoms.” International Journal of Molecular Sciences, vol. 19, no. 12, Dec. 2018, p. 3869. PubMed Central, https://doi.org/10.3390/ijms19123869.
Culjak, Marija, et al. “The Association between TNF-Alpha, IL-1 Alpha and IL-10 with Alzheimer’s Disease.” Current Alzheimer Research, vol. 17, no. 11, 2020, pp. 972–84. PubMed, https://doi.org/10.2174/1567205017666201130092427.
Dhamodharan, Umapathy, et al. “Genetic Association of IL-6, TNF-α and SDF-1 Polymorphisms with Serum Cytokine Levels in Diabetic Foot Ulcer.” Gene, vol. 565, no. 1, July 2015, pp. 62–67. PubMed, https://doi.org/10.1016/j.gene.2015.03.063.
Ding, Cheng, et al. “TNF-α Gene Promoter Polymorphisms Contribute to Periodontitis Susceptibility: Evidence from 46 Studies.” Journal of Clinical Periodontology, vol. 41, no. 8, Aug. 2014, pp. 748–59. PubMed, https://doi.org/10.1111/jcpe.12279.
Driscoll, Conor B., et al. “Long-Term Risk of de Novo Malignancy with Tumor Necrosis Factor Alpha (TNF) Inhibitor Immunosuppression: A Multicenter, Retrospective Cohort Study.” Journal of Inflammation, vol. 22, no. 1, Aug. 2025, p. 34. Springer Link, https://doi.org/10.1186/s12950-025-00445-x.
Durães, Cecília, et al. “Polymorphisms in the TNFA and IL6 Genes Represent Risk Factors for Autoimmune Thyroid Disease.” PloS One, vol. 9, no. 8, 2014, p. e105492. PubMed, https://doi.org/10.1371/journal.pone.0105492.
—. “Polymorphisms in the TNFA and IL6 Genes Represent Risk Factors for Autoimmune Thyroid Disease.” PloS One, vol. 9, no. 8, 2014, p. e105492. PubMed, https://doi.org/10.1371/journal.pone.0105492.
El-Rakabawy, Omar M., et al. “Curcumin Supplementation Improves the Clinical Outcomes of Patients with Diabetes and Atherosclerotic Cardiovascular Risk.” Scientific Reports, vol. 15, no. 1, Aug. 2025, p. 28358. PubMed, https://doi.org/10.1038/s41598-025-09783-5.
Feng, Bo, et al. “Association of Tumor Necrosis Factor α -308G/A and Interleukin-6 -174G/C Gene Polymorphism with Pneumonia-Induced Sepsis.” Journal of Critical Care, vol. 30, no. 5, Oct. 2015, pp. 920–23. PubMed, https://doi.org/10.1016/j.jcrc.2015.04.123.
Frankola, Kathryn A., et al. “Targeting TNF-Alpha to Elucidate and Ameliorate Neuroinflammation in Neurodegenerative Diseases.” CNS & Neurological Disorders Drug Targets, vol. 10, no. 3, May 2011, pp. 391–403. PubMed Central, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4663975/.
Gichohi-Wainaina, Wanjiku N., et al. “Tumour Necrosis Factor Allele Variants and Their Association with the Occurrence and Severity of Malaria in African Children: A Longitudinal Study.” Malaria Journal, vol. 14, June 2015, p. 249. PubMed, https://doi.org/10.1186/s12936-015-0767-3.
Idriss, H. T., and J. H. Naismith. “TNF Alpha and the TNF Receptor Superfamily: Structure-Function Relationship(s).” Microscopy Research and Technique, vol. 50, no. 3, Aug. 2000, pp. 184–95. PubMed, https://doi.org/10.1002/1097-0029(20000801)50:3<184::AID-JEMT2>3.0.CO;2-H.
Jannatifar, Rahil, et al. “Nanomicelle Curcumin Improves Oxidative Stress, Inflammatory Markers, and Assisted Reproductive Techniques Outcomes in Endometriosis Cases: A Randomized Clinical Trial.” Naunyn-Schmiedeberg’s Archives of Pharmacology, vol. 398, no. 9, Sept. 2025, pp. 11933–41. PubMed, https://doi.org/10.1007/s00210-025-03958-7.
Kakino, Satomi, et al. “Pivotal Role of TNF-α in the Development and Progression of Nonalcoholic Fatty Liver Disease in a Murine Model.” Hormone and Metabolic Research = Hormon- Und Stoffwechselforschung = Hormones Et Metabolisme, vol. 50, no. 1, Jan. 2018, pp. 80–87. PubMed, https://doi.org/10.1055/s-0043-118666.
Keramatzadeh, Sara, et al. “Effects of Resveratrol Supplementation on Inflammatory Markers, Fatigue Scale, Fasting Blood Sugar and Lipid Profile in Relapsing-Remitting Multiple Sclerosis Patients: A Double-Blind, Randomized Placebo-Controlled Trial.” Nutritional Neuroscience, vol. 28, no. 7, July 2025, pp. 854–62. PubMed, https://doi.org/10.1080/1028415X.2024.2425649.
Khan, Saif, et al. “TNF-α -308 G > A (Rs1800629) Polymorphism Is Associated with Celiac Disease: A Meta-Analysis of 11 Case-Control Studies.” Scientific Reports, vol. 6, Sept. 2016, p. 32677. PubMed, https://doi.org/10.1038/srep32677.
Locksley, Richard M., et al. “The TNF and TNF Receptor Superfamilies: Integrating Mammalian Biology.” Cell, vol. 104, no. 4, Feb. 2001, pp. 487–501. www.cell.com, https://doi.org/10.1016/S0092-8674(01)00237-9.
Lo, Chih Hung, and Jialiu Zeng. “TNF as a Mediator of Metabolic Inflammation and Body-Brain Interaction in Obesity-Driven Neuroinflammation and Neurodegeneration.” Ageing Research Reviews, vol. 112, Dec. 2025, p. 102891. ScienceDirect, https://doi.org/10.1016/j.arr.2025.102891.
Lo, Chih Hung. “TNF Receptors: Structure-Function Relationships and Therapeutic Targeting Strategies.” Biochimica et Biophysica Acta (BBA) – Biomembranes, vol. 1867, no. 1, Jan. 2025, p. 184394. ScienceDirect, https://doi.org/10.1016/j.bbamem.2024.184394.
Ma, Ke, et al. “Pathogenetic and Therapeutic Applications of Tumor Necrosis Factor-α (TNF-α) in Major Depressive Disorder: A Systematic Review.” International Journal of Molecular Sciences, vol. 17, no. 5, May 2016, p. 733. PubMed Central, https://doi.org/10.3390/ijms17050733.
MacIntyre, Elaina A., et al. “GSTP1 and TNF Gene Variants and Associations between Air Pollution and Incident Childhood Asthma: The Traffic, Asthma and Genetics (TAG) Study.” Environmental Health Perspectives, vol. 122, no. 4, Apr. 2014, pp. 418–24. PubMed, https://doi.org/10.1289/ehp.1307459.
Majumder, Poulami, et al. “Association of Tumor Necrosis Factor-α (TNF-α) Gene Promoter Polymorphisms with Aggressive and Chronic Periodontitis in the Eastern Indian Population.” Bioscience Reports, vol. 38, no. 4, Aug. 2018, p. BSR20171212. PubMed, https://doi.org/10.1042/BSR20171212.
Olszewski, Maciej B., et al. “TNF Trafficking to Human Mast Cell Granules: Mature Chain-Dependent Endocytosis.” Journal of Immunology (Baltimore, Md.: 1950), vol. 178, no. 9, May 2007, pp. 5701–09. PubMed, https://doi.org/10.4049/jimmunol.178.9.5701.
“Pathophysiology of Autoimmune Diseases and Inflammation.” AJMC, https://www.ajmc.com/view/pathophysiology-of-autoimmune-diseases-and-inflammation. Accessed 10 Dec. 2021.
Randall, Louise M., and Christian R. Engwerda. “TNF Family Members and Malaria: Old Observations, New Insights and Future Directions.” Experimental Parasitology, vol. 126, no. 3, Nov. 2010, pp. 326–31. ScienceDirect, https://doi.org/10.1016/j.exppara.2010.04.016.
—. “TNF Family Members and Malaria: Old Observations, New Insights and Future Directions.” Experimental Parasitology, vol. 126, no. 3, Nov. 2010, pp. 326–31. ScienceDirect, https://doi.org/10.1016/j.exppara.2010.04.016.
Reséndiz-Hernández, Juan Manuel, et al. “Identification of Genetic Variants in the TNF Promoter Associated with COPD Secondary to Tobacco Smoking and Its Severity.” International Journal of Chronic Obstructive Pulmonary Disease, vol. 10, 2015, pp. 1241–51. PubMed, https://doi.org/10.2147/COPD.S83298.
Stappers, M. H. T., et al. “Polymorphisms in Cytokine Genes IL6, TNF, IL10, IL17A and IFNG Influence Susceptibility to Complicated Skin and Skin Structure Infections.” European Journal of Clinical Microbiology & Infectious Diseases: Official Publication of the European Society of Clinical Microbiology, vol. 33, no. 12, Dec. 2014, pp. 2267–74. PubMed, https://doi.org/10.1007/s10096-014-2201-0.
Subramanian, Veedamali S., et al. “Tumor Necrosis Factor Alpha Reduces Intestinal Vitamin C Uptake: A Role for NF-ΚB-Mediated Signaling.” American Journal of Physiology – Gastrointestinal and Liver Physiology, vol. 315, no. 2, Aug. 2018, pp. G241–48. PubMed Central, https://doi.org/10.1152/ajpgi.00071.2018.
Tavares, M., et al. “Tumour Necrosis Factor-Alpha (-308G/A) Promoter Polymorphism Is Associated with Ulcerative Colitis in Brazilian Patients.” International Journal of Immunogenetics, vol. 43, no. 6, Dec. 2016, pp. 376–82. PubMed, https://doi.org/10.1111/iji.12289.
Ueda, Mayu, et al. “A Short-Term Treatment with Tumor Necrosis Factor-Alpha Enhances Stem Cell Phenotype of Human Dental Pulp Cells.” Stem Cell Research & Therapy, vol. 5, no. 1, Feb. 2014, p. 31. PubMed Central, https://doi.org/10.1186/scrt420.
Wu, Jun-Cang, et al. “Gene Polymorphisms and Circulating Levels of the TNF-Alpha Are Associated with Ischemic Stroke: A Meta-Analysis Based on 19,873 Individuals.” International Immunopharmacology, vol. 75, Oct. 2019, p. 105827. PubMed, https://doi.org/10.1016/j.intimp.2019.105827.
Yucesoy, Berran, et al. “Genetic Variants in TNFα, TGFB1, PTGS1 and PTGS2 Genes Are Associated with Diisocyanate-Induced Asthma.” Journal of Immunotoxicology, vol. 13, no. 1, 2016, pp. 119–26. PubMed, https://doi.org/10.3109/1547691X.2015.1017061.
Zhang, Guimin, et al. “The Role of TNF Alpha Polymorphism and Expression in Susceptibility to Nasal Polyposis.” Immunological Investigations, vol. 47, no. 4, May 2018, pp. 360–71. Taylor and Francis+NEJM, https://doi.org/10.1080/08820139.2018.1433204.
Zhang, Peng, et al. “Tumor Necrosis Factor-Alpha Gene Polymorphisms and Susceptibility to Ischemic Heart Disease.” Medicine, vol. 96, no. 14, Apr. 2017, p. e6569. PubMed Central, https://doi.org/10.1097/MD.0000000000006569.
—. “Tumor Necrosis Factor-Alpha Gene Polymorphisms and Susceptibility to Ischemic Heart Disease.” Medicine, vol. 96, no. 14, Apr. 2017, p. e6569. PubMed Central, https://doi.org/10.1097/MD.0000000000006569.