Key takeaways:
- Tyramine is a naturally occurring biogenic amine that can cause blood vessels to constrict.
- Tyramine buildup can result in serious symptoms, such as heart palpitations, spiking blood pressure, stroke-like symptoms, nausea, gastrointestinal issues, migraines, and brain fog.
- Genetic variants in three key enzymes (MAOA, FMO3, and CYP2D6) impact how well you break down and eliminate tyramine.
- Medications, especially MAOIs, also impact tyramine metabolism.
What is tyramine intolerance? Symptoms, foods, and breaking it down
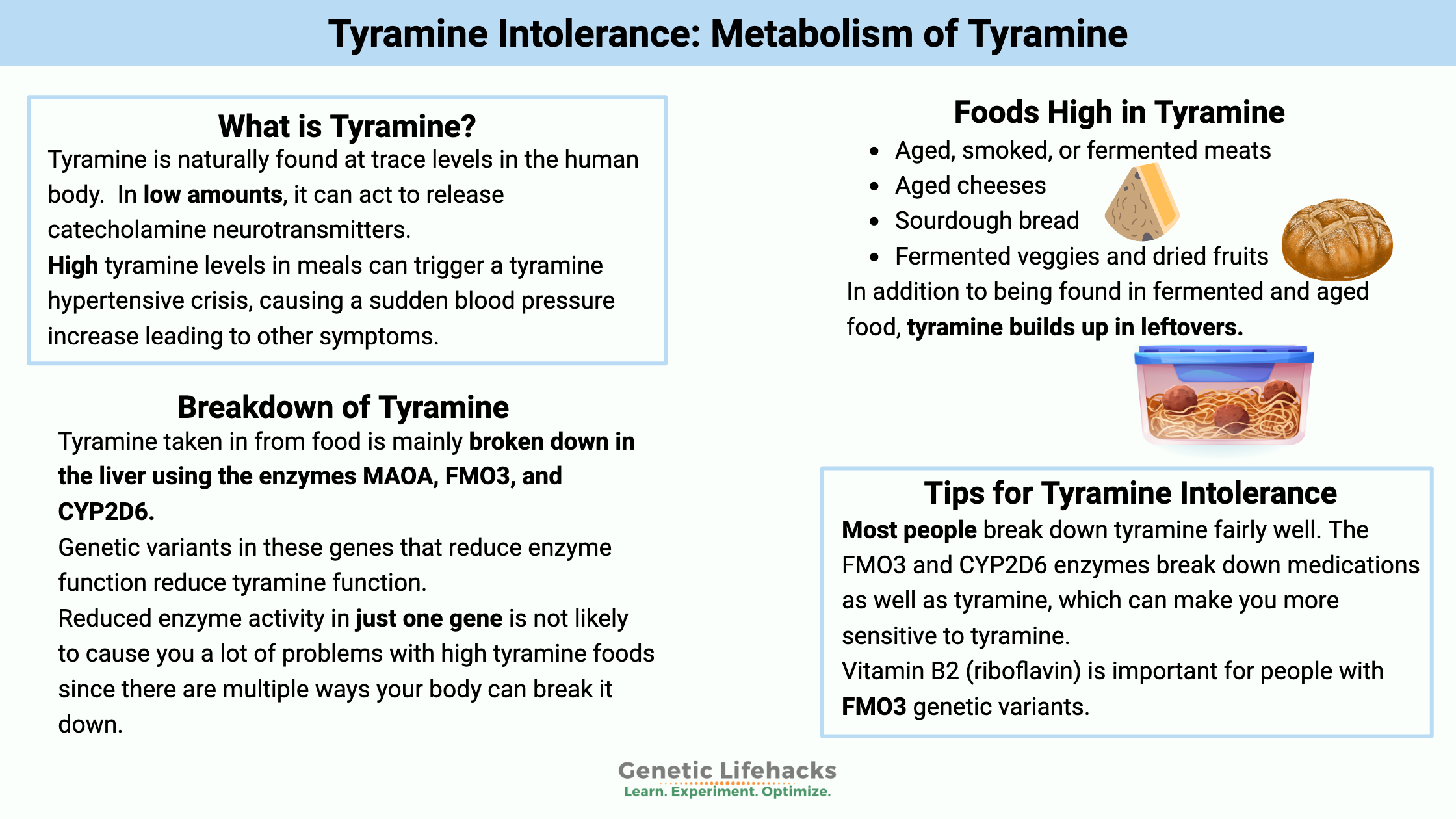
Tyramine is a biogenic amine that is found in high amounts in certain foods and produced in low amounts in the body. It causes the release of norepinephrine in the body, which then increases blood pressure.
Tyramine intolerance is a sensitivity to foods high in tyramine that triggers symptoms like severe migraines, high blood pressure, and hives. It is caused by the body not properly breaking down dietary tyramine, which is often found in aged, fermented, or slightly spoiled foods.
High tyramine levels in meals can trigger a tyramine hypertensive crisis, commonly known as the ‘cheese effect.’ This is usually associated with taking a drug type called an MAO-A inhibitor (MAOI). People on an MAOI are cautioned by their doctor about the dietary interactions, such as with aged cheeses. The hypertensive crisis is caused by too much tyramine, causing a sudden blood pressure increase, leading to other symptoms.
In addition to medication interactions with MAOIs, genetic variants play a role in how well the body handles tyramine. I’ll start with some background on tyramine and then explain the genetic susceptibility.
What is tyramine?
Tyramine is a biogenic amine, which refers to its chemical structure with nitrogen at its base. Other biogenic amines include histamine, spermidine, dopamine, serotonin, epinephrine, and norepinephrine, which are all amines that we create naturally.
It is naturally found at trace levels in the human body. Tyramine, in low amounts, causes the release of catecholamine neurotransmitters. It can cross the blood-brain barrier and act as a neuromodulator in the brain. In the body, it causes the release of norepinephrine, which causes blood vessel constriction.[ref]
Bacteria convert the amino acid tyrosine into tyramine, and fermented foods, aged cheeses, and foods close to spoiling can have very high amounts of tyrosine. This is where the term ‘cheese effect‘ comes into play, because researchers first discovered the hypertensive effects of tyrosine when people on MAOI drugs ate aged cheeses. [ref]
In addition, certain gut microbes can also produce tyramine, adding to the body’s overall tyramine levels.
How does the body get rid of tyramine?
In the intestines, tyramine is absorbed from foods and from gut microbial production. From there, it enters the circulation and is then broken down primarily in the liver.
Tyramine is mainly broken down (metabolized) in the body using these three enzymes:
| Enzyme | Role |
|---|---|
| MAO-A | Primary enzyme, breaks down neurotransmitters; inhibited by MAOIs, increasing risk |
| FMO3 | Supports tyramine breakdown |
| CYP2D6 | Liver enzyme, detoxifies various compounds |
MAO-A is an enzyme that is the primary way the body breaks down tyramine. It also metabolizes (breaks down) several neurotransmitters, including dopamine. Thus, inhibiting MAO-A is one way to increase dopamine levels. Drugs that act as MAO-A inhibitors (MAOIs) can be used as antidepressants, although they usually aren’t the first drug choice due to the interactions with tyramine in foods.
CYP2D6 is a detoxification enzyme produced primarily in the liver and in the brain. It breaks down toxins, medications, and some substances produced in the body, such as serotonin. In tyramine metabolism, the CYP2D6 enzyme acts as a catalyst to convert tyramine to dopamine.[ref]
FMO3 (flavin-containing monooxygenase 3) is another enzyme produced in the liver that can metabolize tyramine. Specifically, FMO3 oxidizes tyramine, converting it into trans-oxime.[ref]
Tyramine reactions:
If you get too much tyramine due to eating foods high in tyramine and not breaking down the tyramine (e.g., when taking an MAO-A inhibitor), it can throw your body into a hypertensive crisis, raising systolic blood pressure by 30 mmHg or more.
This is called the ‘tyramine pressor response’. Tyramine takes the place of other neurotransmitters, triggering the body to release a bunch of norepinephrine, constricting blood vessels and raising blood pressure.[ref]
Interestingly, some of the first studies on the pressor effect of raising blood pressure were done in the early 1900s using rotting horse meat.[ref]
You may wonder why everyone doesn’t have a stroke or heart attack after eating a salami and aged cheese sandwich on sourdough bread. There are a couple of reasons for this:
- First, most people break down tyramine fairly well. There are three different enzyme pathways to take care of it.
- Second, repeated exposure to tyramine will decrease the tyramine pressor response. It’s the change from typically not eating foods high in tyramine to suddenly chowing down on them that can cause a response. For instance, eating a healthy diet full of fresh foods — and then hitting the holiday buffet and having salami, cheese, and olives, chased with a glass of red wine.
Tyramine Sensitivity and Migraines
Research shows that for people susceptible to migraines, the list of foods high in tyramine may correspond to their list of ‘triggers’.
- Many people with either cluster headaches or migraines don’t break down tyramine well.[ref]
- Researchers think that vasoconstriction triggered by tyramine initiates migraines.[ref][ref]
Notable/Interesting Points:
- Tyramine is chemically similar to amphetamines but does not have the same effects.[ref]
- Florida banned tyramine in 2012 as a Schedule I drug. (Does this make aged cheese illegal in Florida?)
Which foods are high in tyramine?
Foods high in tyramine include:
| High Sources | Medium Sources |
|---|---|
| Aged/smoked meats (salami, bacon, jerky) | Olives |
| Aged cheeses (cheddar, Swiss, Parmesan, feta) | Chocolate |
| Sourdough, homemade yeast bread | Snow peas, edamame |
| Marmite, fermented veggies, dried fruits | Avocados, bananas |
| Some beers/wines (esp. unpasteurized) | Pineapple, eggplant |
| Figs, yogurt, sour cream | |
| Peanuts, Brazil nuts | |
| Fava beans |
How do you know if you have histamine intolerance or tyramine intolerance?
Many foods that are high in tyramine are also high in histamine. You may find it difficult to know whether you’re reacting to histamine or tyramine in foods. Read this histamine intolerance article to learn about genetic susceptibility, similarities, and symptom differences.
Tyramine Metabolism Genotype Report
Access this content:
An active subscription is required to access this content.
Lifehacks: Natural solutions for tyramine
Riboflavin for tyramine intolerance:
Vitamin B2 (riboflavin) is important for people with FMO3 genetic variants.[ref][ref] Make sure you get enough riboflavin via your diet or consider supplementing with riboflavin.
This may be one reason that riboflavin works to prevent migraines in some people…
Low tyramine diet (tyramine-containing foods list):
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
Histamine Intolerance
Excess histamine can cause allergy-type reactions in some people.
Serotonin
Your genes play a role in how much serotonin is made, how it is broken down, and how cells receive the serotonin signal.
Your need for riboflavin (B2): MTHFR and other genetic variants
Riboflavin (Vitamin B2) is a water-soluble vitamin that is a cofactor for many enzymes in the body.
Detoxification: Phase I and Phase II Metabolism
Our body has an amazing capacity to rid itself of harmful substances. We take in toxins daily by eating natural plant toxins. We are exposed to toxicants (man-made toxins) through pesticide residue, air pollution, skincare products, and medications.
Trimethylaminuria: Genetic variants that cause a malodorous body odor
Often referred to as ‘fish odor disease’, trimethylaminuria causes a strong odor in sweat, urine, and breath. It is caused by mutations in the FMO3 gene.
References:
“Avoid the Combination of High-Tyramine Foods and MAOIs.” Mayo Clinic, https://www.mayoclinic.org/diseases-conditions/depression/expert-answers/maois/faq-20058035. Accessed 16 Nov. 2022.
Barger, G., and G. S. Walpole. “Isolation of the Pressor Principles of Putrid Meat.” The Journal of Physiology, vol. 38, no. 4, Mar. 1909, pp. 343–52. PubMed Central, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1533642/.
Bushueva, Olga, et al. “The Flavin-Containing Monooxygenase 3 Gene and Essential Hypertension: The Joint Effect of Polymorphism E158K and Cigarette Smoking on Disease Susceptibility.” International Journal of Hypertension, vol. 2014, Aug. 2014, p. e712169. www.hindawi.com, https://doi.org/10.1155/2014/712169.
Cronometer: Eat Smarter. Live Better. https://cronometer.com/. Accessed 16 Nov. 2022.
D’Andrea, G., et al. “Biochemistry of Neuromodulation in Primary Headaches: Focus on Anomalies of Tyrosine Metabolism.” Neurological Sciences, vol. 28, no. 2, May 2007, pp. S94–96. Springer Link, https://doi.org/10.1007/s10072-007-0758-4.
Hotamisligil, G. S., and X. O. Breakefield. “Human Monoamine Oxidase A Gene Determines Levels of Enzyme Activity.” American Journal of Human Genetics, vol. 49, no. 2, Aug. 1991, pp. 383–92.
How a Migraine Happens. 26 Nov. 2019, https://www.hopkinsmedicine.org/health/conditions-and-diseases/headache/how-a-migraine-happens.
Kashyap, A. S., and Surekha Kashyap. “Fish Odour Syndrome.” Postgraduate Medical Journal, vol. 76, no. 895, May 2000, pp. 318–318. pmj.bmj.com, https://doi.org/10.1136/pmj.76.895.318a.
Koukouritaki, Sevasti B., et al. “Discovery of Novel Flavin-Containing Monooxygenase 3 (FMO3) Single Nucleotide Polymorphisms and Functional Analysis of Upstream Haplotype Variants.” Molecular Pharmacology, vol. 68, no. 2, Aug. 2005, pp. 383–92. PubMed, https://doi.org/10.1124/mol.105.012062.
Manning, Nigel J., et al. “Riboflavin-Responsive Trimethylaminuria in a Patient with Homocystinuria on Betaine Therapy.” JIMD Reports, vol. 5, 2012, pp. 71–75. PubMed, https://doi.org/10.1007/8904_2011_99.
Niwa, Toshiro, et al. “Human Liver Enzymes Responsible for Metabolic Elimination of Tyramine; a Vasopressor Agent from Daily Food.” Drug Metabolism Letters, vol. 5, no. 3, Aug. 2011, pp. 216–19. PubMed, https://doi.org/10.2174/187231211796905026.
NM_006894.5(FMO3):C.913G>T (p.Glu305Ter) AND Trimethylaminuria – ClinVar – NCBI. https://www.ncbi.nlm.nih.gov/clinvar/RCV000017697.30/. Accessed 16 Nov. 2022.
NM_001002294.3(FMO3):C.589_590TG[1] (p.Cys197_Asp198delinsTer) AND Trimethylaminuria – ClinVar – NCBI. https://www.ncbi.nlm.nih.gov/clinvar/RCV000490504.1/. Accessed 16 Nov. 2022.
Rafehi, Muhammad, et al. “Highly Variable Pharmacokinetics of Tyramine in Humans and Polymorphisms in OCT1, CYP2D6, and MAO-A.” Frontiers in Pharmacology, vol. 10, Oct. 2019, p. 1297. PubMed Central, https://doi.org/10.3389/fphar.2019.01297.
Sathyanarayana Rao, T. S., and Vikram K. Yeragani. “Hypertensive Crisis and Cheese.” Indian Journal of Psychiatry, vol. 51, no. 1, 2009, pp. 65–66. PubMed Central, https://doi.org/10.4103/0019-5545.44910.
tammy-lrome. “Low-Tyramine Diet Essentials.” Migraine.Com, https://migraine.com/blog/low-tyramine-diet-essentials. Accessed 16 Nov. 2022.
Walker, S. E., et al. “Tyramine Content of Previously Restricted Foods in Monoamine Oxidase Inhibitor Diets.” Journal of Clinical Psychopharmacology, vol. 16, no. 5, Oct. 1996, pp. 383–88. PubMed, https://doi.org/10.1097/00004714-199610000-00007.
Xu, Meijuan, et al. “Genetic and Nongenetic Factors Associated with Protein Abundance of Flavin-Containing Monooxygenase 3 in Human Liver.” The Journal of Pharmacology and Experimental Therapeutics, vol. 363, no. 2, Nov. 2017, pp. 265–74. PubMed, https://doi.org/10.1124/jpet.117.243113.
Xu, Xiaohui, et al. “Association Study between the Monoamine Oxidase A Gene and Attention Deficit Hyperactivity Disorder in Taiwanese Samples.” BMC Psychiatry, vol. 7, Feb. 2007, p. 10. PubMed, https://doi.org/10.1186/1471-244X-7-10.
Zubiaur, Pablo, et al. “SLCO1B1 Phenotype and CYP3A5 Polymorphism Significantly Affect Atorvastatin Bioavailability.” Journal of Personalized Medicine, vol. 11, no. 3, Mar. 2021, p. 204. PubMed, https://doi.org/10.3390/jpm11030204.